

1
LIST OF ABBREVIATIONS
JEE (Main) Joint Entrance Examination (Main)
AICTE All India Council of Technical Education
JoSAA Joint Seat Allocation Authority
CSAB Central Seat Allocation Board
B.E. Bachelor of Engineering
B.Tech. Bachelor of Technology
B.Arch. Bachelor of Architecture
B.Planning Bachelor of Planning
CoA Council of Architecture
MoE Ministry of Education
NTA National Testing Agency
GoI
Government of India
GST Goods & Service Tax
IIT Indian Institute of Technology
IIIT Indian Institute of Information Technology
NIT
National Institute of Technology
DASA Direct Admission for Students Abroad
CFTI Centrally Funded Technical Institute
UFM
Unfair Means
CBT Computer Based Test
NEP National Education Policy
RPwD Rights of Persons with Disabilities
QP
Question Paper
TPC Test Practice Centre
FAQ Frequently Asked Questions
QRS
Query Redressal System
EWS Economically Weaker Section
OBC (NCL) Other Backward Class (Non-Creamy Layer)
SC
Scheduled Castes
ST
Scheduled Tribes
OCI Overseas Citizen of India

2
S. No. Particulars Page
No.
A.
Important Information and Dates at a Glance 4-10
Chapter 1- Introduction
1.1 About National Testing Agency (NTA)
11
Chapter 2 – Joint Entrance Examination (Main) – 2024
2.1 About JEE (Main)
12
Chapter 3 - Examination Scheme
3.1 Mode of Examination
13
3.2 Choice of Medium of Question Papers (QP)
13
3.3 Scheme of Examination 14
3.4 Pattern of Examination 14-18
3.5 Duration of Test 18
3.6 Syllabus of Examination 19
Chapter 4 - Eligibility and Qualifications
4.1 Age Criteria 20
4.2 List of Qualifying Examination (QE) 20
4.3 Year of Appearance in Qualifying Examination (QE) 21
4.4 State of Eligibility 21
Chapter 5 – Reservation
5.1 Reservation
22
5.2 (a) Guidelines for conducting written examination for Persons with Benchmark Disabilities
above 40% vide letter dated 29 August 2018 from the Ministry of Social Justice and
Empowerment
23
5.2 (b) Guidelines for conducting written examination for persons with specified disabilities
covered under the definition of Section 2(s) of the RPwD Act, 2016 but not covered
under the definition of Section 2(r) of the said Act, i.e. persons having less than 40%
disability and having difficulty in writing
24
5.3 State Government Policy for Disability
25
Chapter 6 – Choice of Cities 26
Chapter 7 – Particulars to be Filled in the JEE (Main) – 2024 Online Application Form 27-30
Chapter 8 - Admit Card 31
Chapter 9 – Schedule of Examination
32
Chapter 10 – Important Instructions for the Candidates 33-34
Chapter 11 – Unfair Means Practices and Breach of Examination Rules
11.1 Definition 35
11.2 Punishment for Using Unfair Means Practices 36
11.3 Cancellation of Result 36
Chapter 12 – Display of Answer Key for Challenge
12.1 Display of Answer Key Challenge 37
12.2 Display of Recorded Responses 37

3
Chapter 13 – JEE (Main) NTA Score for B.E./B.Tech. B.Arch. and B.Planning
13.1 JEE (Main) NTA Score for B.E./B.Tech. B.Arch. and B.Planning 38
13.2 Compilation and Display of Result/NTA score of Paper 1(B.E./B.Tech.) 38
13.3 Compilation and Display of Result/NTA score of Paper 2A & 2B (B.Arch. &
B.Planning
.
)
39
13.4 Re-evaluation/Re-checking of Result 40
13.5 Use of Scores of JEE (Main) – 2024 by Other Organizations 40
Chapter 14 – Admission
14.1 Eligibility for Admission to NITs, IIITs, and CFTIs participating through the
Central Seat Allocation Board (CSAB)
41
14.2 Eligibility for Admission to Other Institutions 42
14.3 Seat Allocation Process and Admission Procedure 42
14.4 Eligibility Criteria for Appearing in JEE (Advanced) – 2024 43
Chapter 15 - National Test Abhyas 44
Chapter 16
-
Miscellaneous
16.1 Query Redressal System (QRS) 45
16.2 Correspondence with NTA 45
16.3
Weeding Out Rules
45
16.4 Legal Jurisdiction 45
16.5 RTI 45
S. No. Annexure No.
Particulars Page No.
1. I
Disability Certificate having disability less than 40%
47
2. II
Letter of Undertaking for Using Own Scribe
48
S. No. Appendix No.
Particulars Page No.
1 I List of Examination Cities for JEE (Main) – 2024 49-56
2 II Procedure for Online Payment of Fees and Helpline for Payment-Related
Queries
57-58
3
III
Computer Based Test (CBT)
Guidelines
5
9
-
6
4
4 IV NTA Test Practice Centres (TPCs) 65
5 V Normalization Procedure based on Percentile Score 66-70
6 VI Syllabus of JEE (Main) – 2024 71-91
7 VII Replica of JEE (Main) – 2024 Application Form 92-109

4
IMPORTANT INFORMATION AT A GLANCE
(Please refer to Information Bulletin for details)
1. Important Dates :
EVENTS DATES
(a)
Session-1: JEE (Main) - January 2024
Online Submission of Application Form
01 November 2023 to 30 November 2023
(up to 09:00 P.M.)
Last date for successful transaction of prescribed
A
pplication Fee
30 November 2023 (up to 11:50 P.M.)
Announcement of the City of Examination By the second week of January 2024
Downloading Admit Cards from the NTA website 03 days before the actual date of the
Examination
Dates of Examination Between 24 January and
01 February 2024
Display of Question Paper attempted by the
Candidate and Answer Keys for inviting challenges
To be displayed on the NTA website
Declaration of Result
12 February 2024
(b)
Session-2: JEE (Main) - April 2024
Online Submission of Application Form
02 February 2024 to 02 March 2024
(up to 09:00 P.M.)
Last date for successful transaction of prescribed
Application Fee
02 March 2024
(up to 11:50 P.M.)
Announcement of the City of Examination By the third week of March 2024
Downloading Admit Cards from the NTA website 03 days before the actual date of the
Examination
Dates of Examination Between 01 April and 15 April 2024
Display of Question Paper attempted by the
Candidate and Answer Keys for inviting challenges
To be displayed on the NTA website
Declaration of Result
25 April 2024
(c)
Duration of Examination for each Session of JEE (Main) - 2024 :
Paper 1 (B.E./B.Tech.)
or
Paper 2A (B.Arch.)
or
Paper 2B (B.Planning
.
)
03 Hours
B.Arch. & B.Planning (both) 03 Hours 30 Minutes
(d) Timing of Examination for each Session: JEE (Main) - 2024 :
Duration of Examination First Shift Second Shift
For 3 Hours Paper 09.00 A.M. to 12.00 Noon
(IST)
03.00 P.M. to 06.00 P.M.
(IST)
For 3 Hours 30 Minutes
Paper
09.00 A.M. to 12.30 P.M.
(IST)
03.00 P.M. to 06.30 P.M.
(IST)

5
Fee Payable for JEE (Main) – 2024 for each session:
(Through Credit Card/Debit Card (except Master/Visa Card)/Net Banking/UPI):
Candidates are requested to fill in the Application Form carefully. No corrections will be
permitted once the Application Form is submitted.
Note: Multiple Application Forms submitted by a candidate will not be accepted and will lead
to the cancellation of his/her result.
(i) A candidate has the option to apply for one Session or both Sessions (Session 1 and Session
2 of 2024) together and pay the exam fee accordingly. In other words, if a candidate wishes
to apply only for one Session, he/she has to pay the Examination Fee only for that Session
during the current application period and will have the opportunity to apply again for Session
2 (April 2024).
(ii) If a candidate wishes to apply for Session 2, the candidate can log in and pay the Examination
Fee for Session 2 during that period.
Fee payable for JEE (Main) – 2024
(through Credit Card /Debit Card (except
Master/Visa Card) / Net-Banking/UPI)
Type of
Candidate
Centres in
India
(Fee in ₹)
Centres
Outside
India
(Fee in ₹)
Paper 1: B.E./B. Tech
or
Paper 2A: B. Arch
or
Paper 2B: B.Planning
General
Male 1000 5000
Female 800 4000
Gen-EWS/ OBC
(NCL)
Male 900 4500
Female 800 4000
SC/ST/PwD
Male 500 2500
Female 500 2500
Third Gender
500 3000
Paper 1: B.E./B. Tech &
Paper 2A: B. Arch
or
Paper 1: B.E./B. Tech &
Paper 2B: B. Planning
or
Paper 1: B.E./B.Tech,
Paper 2A: B. Arch &
Paper 2B: B.Planning
or
Paper 2A: B. Arch &
Paper 2B: B.Planning
General/Gen-
EWS/OBC (NCL)
Male 2000 10000
Female 1600 8000
SC/ST/PwD
Male 1000 5000
Female 1000 5000
Third Gender 1000 5000
Processing charges and Goods & Service Taxes (GST) are to be paid by the candidate, as
applicable.
6
(iii)The application window for Session 2 will be re-opened as per the details available in the
Informatin Bulletin and will also be notified separately through a Public Notice.
(iv) The fee can be submitted only online through Net Banking, Credit Card, Debit Card, or UPI
Services. Processing charges and GST as applicable are chargeable from the candidate (in
addition to the examination fee) by the concerned Bank/Payment Gateway Integrator. For
details/procedure, please see Appendix-II.
(v) The Confirmation Page of the online Application Form for each session will be generated
only after the successful payment of the fee by the Candidate. In case the Confirmation Page
is not generated after payment of the fee, then the candidate may have to approach the
concerned Bank/Payment Gateway (in the helpline numbers and email given in Appendix-
II) to ensure the successful payment or to obtain the refund of duplicate /multiple payments.
1.Candidates must read carefully the Instructions (including how to fill up the Application Form
online) given in the Information Bulletin available on the NTA Website. Candidates not
complying with the instructions shall be summarily disqualified.
2.Candidates can apply for JEE (Main) – 2024 Session 1 and Session 2 through the “Online”
mode only. The Application Form in any other mode will not be accepted.
3.Submission of the Application Form could be made by the candidate online through the NTA
website: https:// jeemain.nta.ac.in/
4. Instructions for filling Online Application Form:
Download the Information Bulletin and Replica of the Application Form. Read these
carefully to ensure your eligibility.
Follow the steps given below to Apply Online:
Step 1: Registration Process :
Candidate needs to select any one of the following to register for JEE (Main):
To log in/create a Digi locker account through the NAD portal. Or;
To log in/create an Academic Bank of Credits ID (ABC ID) through the ABC ID. Or;
To log in using an Indian Indian Passport Number. Or;
To log in using a Non-Indian Passport Number. Or;
To log in using a PAN Card Number. Or;
To log in using an Aadhaar Enrollment Number.
i. The candidates are advised to register through Digi Locker/ABC ID.
ii. Those candidates who do not wish to register through the Digi Locker / ABC ID,
need to report early (one hour prior to the entry time) at the examination Centre
on the day of the examination.
Under no circumstances, candidates will be allowed to fill more than one Application
Form. Strict action will be taken, even at a later stage, against such candidates who have
filled more than one Application Form.
7
Candidate needs to select options for Authentication at the Examination Centre.
Authenticate through an Aadhaar Card
Authenticate through Valid Government ID with a Photograph
Please note that for authentication through non-Aadhaar options Candidate needs to
reach the Exam Centre 1 hour earlier than other Candidates to get their Biometrics
recorded at the Exam Centre.
Candidate must write his / her Full Name, the same as printed on the
certificate/marksheet of Class 10 / equivalent Examination otherwise the form will be
rejected.
The candidate should submit the required details while filling the Online Application Form
and is also required to create a PASSWORD, choose Security Question, and enter his/her
Answer.
After successful submission of the personal details, an Application number will be generated
and it will be used to complete the remaining Steps of the Application Form and will also be
required for all future reference/correspondence.
This application number will also be used/referred for both Sessions of JEE (Main) - 2024. For
subsequent logins, the candidate will be able to login directly with the respective system-
generated Application Number and created Password.
Step 2: Application Form:
The Candidates can log in with the system-generated Application Number and pre-created
Password or Digi locker ID / ABC ID and their password for completing the Application
Form including filling up personal details, applying for the Paper, choosing the Examination
Cities, providing the details of Educational Qualifications, and uploading the images and
documents (if any).
Upload Scanned Images of the Candidate’s Photograph, Signature, and PwD Certificate/
UDID Card (Swawlamban Card) (wherever applicable):
i. The recent photograph should be either in colour or black & white with 80% face
(without mask) visible including ears against a white background.
ii. The scanned photograph and signature should be in JPG/JPEG format (clearly legible).
iii. The size of the scanned photograph should be between 10 kb to 200 kb (clearly legible).
iv. The size of the scanned signature should be between 4 kb to 30 kb (clearly legible)
v. The size of the scanned copy of the PwD certificate should be in pdf between 50 kb to 300
kb (clearly legible)

8
[Note: The Candidate has to upload only his/her own photograph, signature, and certificate(s) as
mentioned above (and not of anybody else) in a correct/proper manner, as the facility for
correction may not be given in the future.]
In case, it is found at any time in the future that the Candidate has used/uploaded the
photograph, signature, and certificate(s) of someone else in his/her Application Form/Admit
Card, or he/she has tampered his/her Admit Card/Result/Scorecard, these acts of the
candidate shall be treated under Unfair Means (UFM) Practices and actions taken as
detailed under the provisions of the Information Bulletin relating to Unfair Means
Practices.
Please check your photograph and signature before submission of the Application Form. In
case the photograph or signature is blurred or not visible to identify the identity of the
candidate then, the application will be rejected and no option for correction or revision will
be permitted
Step 3: Fee Payment:
After completing Step 1 and Step 2, the candidates have to pay the requisite examination fee.
The fee can be only submitted online through Net Banking, Credit Card, Debit Card, and UPI.
Processing charges and GST as applicable are chargeable to the candidate (in addition to the
examination fee) by the concerned Bank.
The Confirmation Page of the online Application Form will be generated only after successful
payment by the Candidate. In case the Confirmation Page is not generated after payment of fee,
then the candidate may have to approach the concerned Bank (in the helpline number and e-
mail given in the Information Bulletin) for ensuring the successful payment or for obtaining the
refund of duplicate/multiple payments.
Download, save, and print a copy of the Confirmation Page of the Application Form (which
would be downloadable only after the successful remittance of the fee) for future reference.
All 3 Steps can be done together or at separate times. The submission of the Application of a
candidate could be considered successful and his/her candidature would be confirmed only on
the successful transaction/receipt of the prescribed application fee from him/her.
5. Candidates shall ensure that the information entered by them in their respective online
Application Forms is correct.
6. All candidates must ensure that they have provided the correct e-mail address and mobile
number. A copy of the Confirmation Page and Final Score Card of JEE (Main) – 2024 will
also be sent to the registered e-mail address of the Candidate as well as the Parent/Guardian.
7. Information provided by the candidates in their respective online Application Forms, like, the
name of the candidate, contact details, address details, category, PW status, educational
qualification details, date of birth, choice of examination cities, etc. will be treated as final. Any
request for change in such particulars will not be considered by NTA under any
circumstances.
8. NTA does not edit/modify/alter any information entered by the candidates after completion of
the application process under any circumstances. Any request for change in information
thereafter will not be entertained. Therefore, candidates are advised to exercise utmost caution
before filling up the correct details in the Application Form.
9
9. NTA disclaims any liability that may arise to a candidate(s) due to incorrect information
provided by him/her in his/her online Application Form.
10. Candidates must ensure that their email address and mobile number to be registered in their
online Application Form are their own, as relevant/important information/ communication will
be sent by NTA through e-mail on the registered mail address and/or through SMS on the
registered mobile number only. NTA shall not be responsible for any non-communication
/miscommunication with a candidate in the email address or mobile number given by him/her
other than his/her own. Candidates are advised to visit the NTA Website and check their e-
mails regularly for the latest updates.
11. Candidates shall appear at their own cost at the Examination Centre on the date, shift, and
time indicated on their Admit Cards issued by the NTA in due course through its website.
Note:
i. The final submission of the Online Application Form will remain incomplete if Step 3 is not
complete. Such forms will stand rejected and no correspondence on this account will be
entertained.
ii. The entire application process for JEE (Main) - 2024 is online, including uploading scanned
images/documents, payment of fees, and printing of Confirmation Page, Admit Card, etc.
Therefore, candidates are not required to send/submit any document(s) including the
Confirmation Page to NTA through Post/ Fax/WhatsApp/Email/by Hand.
iii. NTA has facilitated all candidates with an additional platform of UMANG and DigiLocker
to download their documents like Confirmation Page, Admit Card, Score Cards, etc.
Instruction will be provided in subsequent phases. Candidates are advised to visit the NTA
Website and check their e-mails regularly for the latest updates.
iv. Usage of Data and Information: NTA/Government of India can use the data provided by the
End Users (test taker in this case) for internal purpose(s) including training, research and
development, analysis, and other permissible purposes (s). However, this information is not
for use by any third party or private agency for any other use.
v.
vi. Candidates are advised to fill only one Application Form and to take utmost care while
filling out the mobile number and e-mail address. Candidates are also advised to use the
mobile number and email address that are accessible to them. Only one mobile number
and email address should be used for one Application Form.
vii. It is mandatory to provide the Mobile Number and e-mail of the Parent or Guardian during
the submission of the online Application Form of JEE (Main) – 2024 as a copy of the
Confirmation Page, The Final Score Card will also be sent to them.
Choice of examination cities displayed to the candidates will be based on the permanent
and correspondence addresses filled during Online Application Form of JEE (Main) –
2024
.

10
Brief Advisory:
Candidates are advised to carry only the following with them into the examination
venue:
i. Admit card along with Self Declaration (Undertaking) downloaded from the
NTA Website (a clear printout on A4 size paper) duly filled in.
ii. A simple transparent Ball Point Pen.
iii.
Additional photograph, to be pasted on the attendance sheet.
iv. Personal transparent water bottle.
v. Sugar tablets/fruits (like banana/apple/orange) in case the candidate is
diabetic.
Note:
1. All Candidates shall ensure that the information (like his/her name, mother’s name,
father’s name, gender, date of birth, category, PwD status, mobile number, e-mail
address, photograph, and signature, choice of cities for examination Centre, etc.) provided
by them in their online Application Form is correct. Candidates are advised to exercise
the utmost care in filling up the correct details in the Online Application Form. Any
request for change in the particulars and uploaded scanned images at any stage will
not be considered by NTA under any circumstances. NTA will not entertain the
corrections sent by the candidate through Post/Fax/WhatsApp/E-mail/by Hand.
2. In case it is found at any time in the future that the Candidate has used/uploaded the
photograph, signature, and certificate(s) of someone else in his/ her Application
Form/Admit Card or he/she has tampered with his/her Admit Card/result, it would be
treated as Unfair Means (UFM) Practices on his/her part and the actions will be taken
under the provisions of Unfair Means Practices, as detailed in the Information Bulletin.
3. Candidates are NOT allowed to carry Instruments, Geometry or Pencil box,
Handbag, Purse, any kind of Paper/ Stationery/ Textual material (printed or written
material), Eatables and Water (loose or packed), Mobile Phone/ Earphone/
Microphone/ Pager, Calculator, DocuPen, Slide Rules, Log Tables, Camera, Tape
Recorder, Electronic
Watches with facilities of calculator, any metallic item or electronic gadgets/ devices
in the Examination Hall/Room.
4. Candidates are requested to retain a copy of the Confirmation Page, Admit Card, and
Scorecard of the JEE (Main) – 2024 as the same will not be available after 31 July 2024.

11
CHAPTER 1
INTRODUCTION
1.1 About National Testing Agency (NTA)
The Ministry of Education (MoE), Government of India (GoI) established the National Testing
Agency (NTA) as an independent, autonomous, and self-sustained premier testing organization under
the Societies Registration Act (1860) for conducting efficient, transparent, and international
standardized tests in order to assess the competency of candidates for admission to premier higher
education institutions with a mission to improve equity and quality in education by developing and
administering research-based valid, reliable, efficient, transparent, fair and international level
assessments.
NTA has created a system that is promoting teaching (by teachers), learning (by students), and
assessment (by parents and institutions). NTA strongly believes in the quality, efficiency,
effectiveness, equity, and security of assessments. To practice these values, NTA is constantly
engaging with its stakeholders, viz. students, parents, teachers, experts, and partner institutions.
The objectives of NTA, inter alia, include:
1. To conduct efficient, transparent, and international standardized tests in order to assess the
competency of candidates for admission.
2. To undertake research on educational, professional, and testing systems to identify gaps in the
knowledge systems and take steps for bridging them.
3. To produce and disseminate information and research on education and professional development
standards.
The Department of Higher Education, Ministry of Education, Government of India has entrusted the
responsibility of conducting the Joint Entrance Examination [JEE (Main)] to the NTA from 2019
onwards.

12
CHAPTER-2
JOINT ENTRANCE EXAMINATION (MAIN) - 2024
2.1 About JEE (Main) - 2024
The Joint Entrance Examination, JEE (Main) comprises two papers. Paper 1 is conducted for
admission to Undergraduate Engineering Programs (B.E/B.Tech.) at NITs, IIITs, other
Centrally Funded Technical Institutions (CFTIs), and Institutions/Universities
funded/recognized by participating State Governments. JEE (Main) is also an eligibility test for
JEE (Advanced), which is conducted for admission to IITs. Paper 2 is conducted for admission
to B. Arch and B. Planning courses in the country.
The JEE (Main) - 2024 is being conducted in 02 (two) sessions for admissions in the next
academic session. The candidates will thus benefit in the following ways:
This will give the candidates two opportunities to improve their scores in the examination
if they are not able to give their best in the first attempt.
In the first attempt, the students will get a first-hand experience of taking an examination
and will know their mistakes which they can improve while attempting for the second time.
This will reduce the chances of dropping a year and droppers would not have to waste an
entire year.
If anyone misses the examination due to reasons beyond control (such as the Board
examination), then he/she will not have to wait for one entire year.
A candidate need not appear in both Sessions. However, if a candidate appears in more
than one Session then his/her best of the JEE (Main) - 2024 NTA Scores will be considered
for preparation of the Merit List/ Ranking.
1. JEE (Main) - 2024 Session 1 for Paper 1 (B.E./B.Tech.) will be held between 24
January and 01 February 2024 and Session 2 will be held between 01 April and 15
April 2024. This is being done to ensure that the JEE (Main) - 2024 does not interfere
with the Board examinations, which may be held at different times across the
States/UTs. Paper 2A and Paper 2B (B. Arch and B. Planning) are also being held
twice a year (January and April 2024).
2. It has been decided to provide choices in one section of each subject of Paper 1 and
Part-I of Paper 2A and 2B to cater to the decision of different Boards across the country
regarding the reduction of the syllabus. However, the total number of questions to be
attempted will remain the same (Physics – 25, Chemistry – 25, and Mathematics – 25),
wherever applicable.

13
CHAPTER 3
EXAMINATION SCHEME
3.1 Mode of Examination
JEE (Main) - 2024 is being conducted in the following modes:
a) Paper 1 (B.E. /B. Tech.) in “Computer Based Test (CBT)” mode only.
b) Paper 2A (B. Arch): Mathematics (Part-I) and Aptitude Test (Part-II) in “Computer Based
Test (CBT)” mode only and Drawing Test (Part-III) in pen and paper (offline) mode, to be
attempted on drawing sheet of A4 size.
c) Paper 2B (B. Planning): Mathematics (Part-I), Aptitude Test (Part-II), and Planning-Based
Questions (Part-III) in Computer-Based Test (CBT) mode only.
3.2 Choice of Medium of Question Papers
Medium of the Question Papers: Drawing from the National Education Policy (NEP), the JEE
(Main) - 2024 is being conducted in English, Hindi, Assamese, Bengali, Gujarati, Kannada,
Malayalam, Marathi, Odia, Punjabi, Tamil, Telugu, and Urdu.
S. No.
Language Examination Centres
1.
English All Examination Centres
2.
English and Hindi All Examination Centres in India
3.
English and Assamese Examination Centres in Assam
4.
English and Bengali Examination Centres in West Bengal, Tripura, and
Andaman & Nicobar Islands
5.
English and Gujarati Examination Centres in Gujarat, Daman & Diu, Dadra
& Nagar Haveli
6.
English and Kannada Examination Centres in Karnataka
7.
English and Malayalam Examination Centres in Kerala and Lakshadweep
8.
English and Marathi Examination Centres in Maharashtra
9.
English and Odia Examination Centres in Odisha
10.
English and Punjabi Examination Centres in Punjab, Chandigarh, and
Delhi/New Delhi (including Faridabad, Ghaziabad,
Gurugram,
Meerut, Noida/Greater Noida
)
11.
English and Tamil Examination Centres in Tamil Nadu, Puducherry, and
Andaman
&
Nicobar Island
s
12.
English and Telugu Examination Centres in Andhra Pradesh and Telangana
13.
English and Urdu All Examination Centres in India

14
The option of language for Question Paper should be exercised carefully while filling up the
Application Form online and it cannot be changed at a later stage.
Please note that for the correctness of the questions in all the question papers, the English
version will be taken as final.
Candidates while attempting the QP must choose the same medium as opted while
registering in the Application form.
3.3 Scheme of Examination
Subject combinations for each paper, type of questions in each paper, and mode of examination are
given in the table below:
PAPER SUBJECTS TYPE OF QUESTIONS MODE OF
EXAMINATION
Paper 1:
B.E./B.Tech.
Mathematics,
Physics and
Chemistry
Objective Type - Multiple
Choice Questions (MCQs) and
Questions for which the answer
is a numerical value, with equal
weightage to Mathematics,
Physics, and Chemistry
“Computer Based Test
(CBT)” mode only
Paper 2A:
B. Arch
Part-I:
Mathematics
Objective Type - Multiple
Choice Questions (MCQs) and
Questions for which the answer
is a numerical value
“Computer Based Test
(CBT)” mode only
Part-II:
Aptitude Test
Objective Type - Multiple
Choice Questions (MCQs)
Part-III: Drawing
Test
Questions to test drawing
aptitude
“Pen and Paper Based”
(offline) mode to be
attempted on a Drawing
sheet
of
A4 size
.
Paper 2B:
B. Planning
Part-I:
Mathematics
Objective Type - Multiple
Choice Questions (MCQs) and
Questions for which the answer
is a numerical value
“Computer Based Test
(CBT)” mode only
Part-II:
Aptitude Test
Objective Type - Multiple
Choice Questions (MCQs)
Part-III:
Planning-Based
Questions
Objective Type - Multiple
Choice Questions (MCQs)
3.4 Pattern of Examination
Paper1: B.E./ B. Tech. in Computer Based Test (CBT) mode:
1. Subject-wise
distribution of
Questions, Total
Number of Questions,
and Marks
Subject Section A Section B Marks
Mathematics
20
*
10*
100
Physics 20* 10* 100
Chemistry
20
*
10*
100
Total 90 300

15
*Each Subject will have two sections. Section A will be of Multiple-Choice Questions (MCQs)
and Section B will contain Questions whose answers are to be filled in as a numerical value.
In Section B, candidates have to attempt any 05 (five) questions out of 10. There will be
negative marking for both Section A and Section B. For each question in Section B, a
candidate has to enter the correct integer value of the answer using the mouse and the on-
screen virtual numeric keypad in the place designated to enter the answer. For Section B, the
answer should be rounded off to the nearest integer.
2.
Marking Scheme for
MCQs
Correct Answer or the Most
Appropriate
Answer
Four marks (+4)
Incorrect Answer
Minus one mark (-1)
Unanswered / Marked for Review
No mark (0)
3.
Marking Scheme for
questions for which the
answer is a Numerical
value
Correct Answer
Four marks (+4)
Incorrect Answer Minus one mark (-1)
Unanswered / Marked for Review
No mark (0)
4.
Method of determining
merit
Conversion of the raw score in Mathematics, Physics, and Chemistry, and
the conversion of the total into NTA scores.
Overall merit shall be prepared by merging NTA scores of all
shifts of all days.
5.
Method of resolving ties
Tie between candidates obtaining equal Total NTA scores in Paper
1: B.E./B.Tech will be resolved in the following manner in
descending order:
1. NTA score in Mathematics, followed by
2. NTA score in Physics, followed by
3. NTA score in Chemistry, followed by
4. Candidate with less proportion of a number of attempted
incorrect answers and correct answers in all the subjects in the
Test, followed by
5. Candidate with less proportion of a number of attempted
incorrect answers and correct answers in Mathematics in the
Test, followed by
6. Candidate with less proportion of a number of attempted
incorrect answers and correct answers in Physics in the Test,
followed by
7. Candidate with less proportion of a number of attempted
incorrect answers and correct answers in Chemistry in the Test
followed by
8. Older in Age followed by
9. Application Number in ascending order
Paper 2A (B. Arch): Mathematics (Part-I) and Aptitude Test (Part-II) in Computer Based
Test (CBT) mode only and Drawing Test (Part-III) in Pen and Paper Based (offline) mode, to
be attempted on a drawing sheet of A4 size.
1.
Subject-wise
distribution of
Questions, Total
Subject Number of Questions Mar
ks
Part I: Mathematics
20* (Section
A
) and 10* (Section
B
)
100
16
Number of Questions,
and Marks
Part II: Aptitude Test 50 200
Part III: Drawing Test
02
100
Total 82
400
*20 questions will be MCQs and 05 (five) questions will have answers to be filled as a
numerical value. There will be negative marking for both Section A and Section B. For each
question in Section B, a candidate has to enter the correct integer value of the answer using
the mouse and the on-screen virtual numeric keypad in the place designated to enter the answer.
The answer should be rounded off to the nearest integer.
2.
Marking Scheme for
MCQs
Correct Answer or the most appropriate
Answer
Four marks (+4)
Incorrect Answer/Multiple Answer
Minus one mark (-1)
Unanswered /Marked for Review
No mark (0)
3.
(a) Marking Scheme for
questions for which
the answer is a
Numerical value
Correct Answer or the most appropriate
Answer
Four marks (+4)
Incorrect Answer
Minus one mark (-1)
Unanswered / Marked for Review
No mark (0)
(b) Marking Scheme for
Drawing Test
(Part III)
Two questions are to be evaluated out of 100 marks.
4.
Method of determining
merit
Conversion of the raw score in Mathematics, Aptitude Test, Drawing
Test, and Total into NTA Scores.
Overall merit shall be prepared by merging the NTA Scores of both
shifts of all days.
5.
Method of resolving
ties
The tie between candidates obtaining equal Total NTA scores in
Paper 2A: B. Arch will be resolved in the following manner:
1. NTA score in Mathematics, followed by
2. NTA score in Aptitude Test, followed by
3. NTA score in Drawing Test, followed by
4. Candidate with less proportion of a number of attempted
incorrect answers and correct answers in all the subjects in the
Test, followed by
5. Candidate with less proportion of a number of attempted
incorrect answers and correct answers in Mathematics (Part-I)
in the Test, followed by
6. Candidate with less proportion of a number of attempted
incorrect answers and correct answers in Aptitude Test (Part-
II)
in the Test followed by
7. Older in Age followed by
8. Application Number in ascending order
Paper 2B (B. Planning) Part-I: Mathematics, Part-II: Aptitude Test, and Part-III: Planning-
Based Questions in Computer-Based Test (CBT) mode only
1.
Subject-wise
distribution of
Questions, Total
Subject Number of Questions Mar
ks
Part-I: Mathematics 20* (Section A) and 10* (Section B)
100

17
Number of Questions,
and Marks
Part-II: Aptitude Test 50 200
Part
-
III: Planning
25
100
Total 105 400
*20 questions will be MCQs and 05 (five) questions will have answers to be filled as a numerical
value. There will be negative marking for both Section A and Section B. For each question in
Section B, enter the correct integer value of the answer using the mouse and the on-screen
virtual numeric keypad in the place designated to enter the answer. The answer should be
rounded off to the nearest integer.
2.
Marking Scheme for
MCQs
Correct Answer or the most appropriate
Answer
Four marks (+4)
Incorrect Answer/Multiple Answer
Minus one mark (-1)
Unanswered /Marked for Review
No mark (0)
3.
Marking Scheme for
questions for which
the answer is a
Numerical value
Correct Answer or the most appropriate
Answer
Four marks (+4)
Incorrect Answer Minus one mark (-1)
Unanswered / Marked for Review No mark (0)
4.
Method of determining
merit
Conversion of the raw score in Mathematics, Aptitude Test, Planning
Based Test, and Total into NTA Scores.
Overall merit shall be prepared by merging NTA Scores of all
shifts of all days.
5.
Method of resolving
ties
Tie between candidates obtaining equal Total NTA scores in B.
Planning will be resolved in the following manner:
1. NTA score in Mathematics, followed by
2. NTA score in Aptitude Test, followed by
3. NTA score in Planning Based Questions, followed by
4. Candidate with less proportion of a number of attempted
incorrect answers and correct answers in all the subjects in the
Test, followed by
5. Candidate with less proportion of a number of attempted
incorrect answers and correct answers in Mathematics (Part-
I) in the Test, followed by
6. Candidate with less proportion of a number of attempted
incorrect answers and correct answers in Aptitude Test (Part-
II) in the Test, followed by
7. Candidate with less proportion of a number of attempted
incorrect answers and correct answers in Planning Based
Questions (Part-III) in the Test followed by
8. Older in Age followed by
9.
Application Number in ascending order
Important Note:
(a) For Multiple Choice Questions: To answer a question, the candidates need to choose one option
corresponding to the correct answer or the most appropriate answer. However, if any anomaly or
discrepancy is found after the process of challenges of the key verification, it shall be addressed in
the following manner:

18
(i) Correct answer or the most appropriate answer: Four marks (+4)
(ii) Any incorrect option marked will be given minus one mark (-1).
(iii) Unanswered/Marked for Review will be given no mark (0).
(iv) If more than one option is found to be correct then Four marks (+4) will be awarded to only
those who have marked any of the correct options.
(v) If all options are found to be correct then Four marks (+4) will be awarded to all those who
have attempted the question.
(vi) If none of the options is found correct or a Question is found to be wrong or a Question is
dropped then full marks will be awarded to all candidates who have appeared
irrespective of the fact whether the question has been attempted or not attempted by the
candidate.
(b) For Numerical Value Questions:
There will be negative marking for Section B. However, if any anomaly or discrepancy is found
after the process of challenges of the key verification, it shall be addressed in the following manner:
i. Correct Answer: Four marks (+4)
ii. Incorrect Answer: Minus one mark (-1)
iii. Unanswered/Marked for Review: No mark (0).
iv. If a question is found to be incorrect or the Question is dropped then Four marks (+4)
will be awarded to all those who have attempted the question. The reason could be due
to human error or technical error.
v. Candidates are advised to do the calculations with the constants given (if any) in the
questions. The answer should be rounded off to the nearest integer.
3.5 Duration of Test
The total time duration for Paper 1 (B.E. /B.Tech.) is given in the table below:
Paper Subjects Total Time Duration
For Non-PwD
Candidates
*For PwD
Candidates
Paper 1
(B.E./B.Tech.)
Physics,
Chemistry, and
Mathematics
03 hours 04 hours
*Note: - Compensatory time for PwD Candidates of 20 minutes for a one-hour
examination
19
Total time duration for Paper 2A (B. Arch), Paper 2B (B.Planning), and Paper 2A (B. Arch)
& Paper 2B (B.Planning) both are given in the table below:
3.6 Syllabus of Examination
The details of the syllabus are given on the website: https://jeemain.nta.ac.in/
Moreover, to cater to the decision of different Boards across the country regarding the reduction
in the syllabus, the NTA has decided to provide a choice in one Section of each Subject of Paper
1 and Part I of Paper 2A and 2B. However, the total number of questions to be attempted will
remain the same (Physics -25, Chemistry – 25, and Mathematics – 25), wherever applicable.
(Refer to Appendix-VI for details).
Paper Subjects
Total Time Duration
For Non-PwD
Candidates
*For PwD
Candidates
Paper 2A
(B.Arch) Only
Mathematics,
Aptitude Test, and
Drawing Test
03 hours 04 hours
Paper 2B
(B.Planning) Only
Mathematics,
Aptitude Test, and
Planning Based
Questions
03 hours 04 hours
Paper 2A (B.Arch)
and Paper 2B
(B.Planning) both
Mathematics,
Aptitude Test,
Drawing Test, and
Planning Based
Questions
03 hours 30
minutes
04 hours 10 minutes
*Note: - Compensatory time for PwD Candidates is 20 minutes for one-hour
examination

20
CHAPTER 4
ELIGIBILITY AND QUALIFICATIONS
4.1 Age Criteria
For appearing in the JEE (Main) - 2024, there is no age limit for the candidates. The candidates
who have passed the class 12/equivalent examination in 2022, 2023, or appearing in 2024
irrespective of their age can appear in JEE (Main) - 2024 examination. However, the candidates
may be required to fulfill the age criteria of the Institute(s) to which they are desirous of taking
admission.
4.2 List of Qualifying Examinations (QE)
i. The final examination of the 10+2 system, conducted by any recognized Central/ State
Board, such as the Central Board of Secondary Education, New Delhi; Council for the Indian
School Certificate Examinations, New Delhi; etc.
ii. Intermediate or two-year Pre-University examination conducted by a recognized Board/
University.
iii. Final examination of the two-year course of the Joint Services Wing of the National
Defense Academy
iv. Senior Secondary School Examination conducted by the National Institute of Open
Schooling with a minimum of five subjects.
v. Any Public School/ Board/ University examination in India or any foreign country is
recognized as equivalent to the 10+2 system by the Association of Indian Universities
(AIU).
vi. Higher Secondary Certificate Vocational Examination.
vii. A Diploma recognized by AICTE or a State board of technical education of at least 3
years duration.
viii. General Certificate Education
(
GCE) examination (London/Cambridge/Sri Lanka) at the
Advanced (A) level.
ix. High School Certificate Examination of the Cambridge University or International
Baccalaureate Diploma of the International Baccalaureate Office, Geneva.
x. Candidates who have completed the Class 12 (or equivalent) examination outside India or
from a Board not specified above should produce a certificate from the Association of
Indian Universities (AIU) to the effect that the examination they have passed is equivalent
to the Class 12 Examination.
xi. In case the Class 12 Examination is not a public examination, the candidate must have passed
at least one public (Board or Pre-University) examination earlier.

21
4.3 Year of Appearance in Qualifying Examination
Only those candidates who have passed the Class 12/equivalent examination in 2022, 2023, or
those who are appearing in the Class 12/equivalent examination in 2024, are eligible to appear
in JEE (Main) - 2024.
Candidates who passed the Class 12/equivalent examination in 2021 or before as well as those
who will appear in such examination in 2024 or later are not eligible to appear in JEE (Main) -
2024.
4.4 State of Eligibility
State code of eligibility means the code of the State from where the candidate has passed the Class
12 (or equivalent) qualifying examination by virtue of which the candidate becomes eligible to
appear in JEE (Main) - 2024. It is important to note that the State code of eligibility does NOT
depend upon the native place or the place of residence of the candidate. For example, if a
candidate appears for the Class 12 (or equivalent) qualifying examination from an Institution
situated in New Delhi and is a resident of Noida, Uttar Pradesh, then the candidate’s State code
of eligibility will be Delhi and NOT Uttar Pradesh.
If a candidate has passed the Class 12 (or equivalent) qualifying examination from one State but
appeared for improvement from another State, the candidate’s State code of eligibility will be
from where the candidate first passed the Class 12 (or equivalent) examination and NOT the State
from where the candidate has appeared for improvement.
Candidates passed/appearing for Class 12 from NIOS should select the State of Eligibility
according to the State in which the study Centre is located.
For Indian nationals passing the Class 12 (or equivalent) examination from Nepal/Bhutan, the
State code of eligibility will be determined based on a permanent address in India as given in the
passport of the candidate.
The State code of eligibility for OCI passing the Class 12 (or equivalent) examination in India is
at par with Indian nationals. However, OCI passing the Class 12 (or equivalent) examination from
an institution abroad are eligible for Other State quota seats or All India quota seats (but NOT for
Home State quota seats) in all NITs, IIITs, and Other-CFTIs.

22
CHAPTER 5
RESERVATIONS
5.1 Indian nationals belonging to certain categories are admitted under the seats reserved for them
in accordance with the rules prescribed by the Government of India. The categories and the
extent of reservation are as follows:
General category belonging to Economically Weaker Section (GEN- EWS) – 10% of seats
in every course. The benefit of reservation will be given only to those General category
candidates who satisfy the conditions given in the OM No. 20013/01/2018-BC-II dated 17
January 2019, issued by the Ministry of Social Justice and Empowerment. The criteria for
GEN-EWS will be as per the prevailing norms and/or notifications of the Government of
India.
Other Backward Classes belonging to the Non-Creamy Layer (OBC- NCL) – 27% of seats
in every course.
i. OBCs should be listed in the current updated central list of OBCs (http://www.ncbc.ac.in).
ii. OBCs present in the State list but not covered in the central list of OBCs (as per the list in
http://www.ncbc.ac.in) are NOT eligible to claim the reservation.
iii. The criteria for OBC-NCL will be as per the notification of the Government of India.
iv. Candidates belonging to the creamy layer of OBC are NOT entitled to reservation. Such
candidates are treated as belonging to the general (GEN), i.e. unreserved category, and they
will be eligible only for the OPEN seats – the seats for which all candidates are eligible.
Scheduled Caste (SC) – 15% of seats in every course.
Scheduled Tribe (ST) – 7.5% of seats in every course.
Persons with Disability (PwD) – 5% seats in each of the GENERAL, GEN-EWS, OBC-
NCL, SC, and ST category seats.
i. Candidates with at least 40% impairment irrespective of the type of disability shall be eligible
for the benefits of the PwD category.
ii. Leprosy-cured candidates who are otherwise fit to pursue the courses are also included in
this category.
iii. Candidates with less than 40% disability, but difficulty in writing, as prescribed by the
Competent authority, are also included in this category.

23
5.2 (a) Guidelines for conducting written examination for Persons with Benchmark
Disabilities above 40% vide letter dated 29 August 2018 from Ministry of Social Justice
and Empowerment
Provisions relating to Persons with Disability (PwD):
As per Section 2(t) of the RPwD Act, “Persons with Disability (PwD)” means a person with
long-term physical, mental, intellectual, or sensory impairment which, in interaction with
barriers, hinders his full and effective participation in society equally with others.
According to Section 2(r) of the RPwD Act, 2016, “persons with benchmark disabilities”
means a person with not less than forty percent (40%) of a specified disability where
specified disability has not been defined in measurable terms and includes a person with
disability where specified disability has been defined in measurable terms, as certified by the
certifying authority.
Facilities for PwD candidates to appear in the exam
As per the guidelines issued by the Department of Empowerment of Persons with Disabilities
(Divyangjan) under the Ministry of Social Justice and Empowerment issued from time to time
on the subject: “Written Examination for Persons with Benchmark Disabilities”, for the
candidate with one of the benchmark disabilities [as defined in Section 2(r) of RPwD Act, 2016],
holding a Disability Certificate in the prescribed format in the Rights of Person with Disabilities
Rules, 2017 (link:
https://upload.indiacode.ac.in/showfile?actid=AC_CEN_25_54_00002_201649_15178073
28299&type=rule&filename=Rules_notified_15.06.pdf ).
a. The facility of a Scribe, in case he/she has a physical limitation and a scribe is essential to
write the examination on his/her behalf, being so certified in the aforesaid format by a
CMO/Civil Surgeon/ Medical Superintendent of a Government Health Care Institution.
b. Compensatory time of one hour for examination of three hours duration, whether such
candidate uses the facility of Scribe or not.
Services of a Scribe
As per the office memorandum of the Ministry of Social Justice and Empowerment (Reference:
F.No. 34-02/2015-DD-III dated August 29, 2018), the PwD candidates who are visually
impaired OR have a disability in the upper limbs OR have lost fingers/hands thereby preventing
them from properly operating the Computer Based Test platform may avail the services of a
scribe (amanuensis).
The scribe will help the Candidate in reading the questions and/or keying in the answers as per
the directions of the Candidate. A scribe will NEITHER explain the questions NOR suggest any
solutions.
24
PwD candidates who desire to avail the services of a scribe need to opt for this during the online
registration of JEE (Main) – 2024. Annexure V
It is to be noted that the Scribe will be provided by the National Testing Agency (NTA) if
requested.
If it is found at any stage that a candidate has availed the services of a scribe and/or availed
the compensatory time but does not possess the extent of disability that warrants the use of a
scribe and/or grant of compensatory time, the candidate will be excluded from the process of
evaluation, ranking, counselling, and admission. In case such a candidate has already been
admitted to any Institution, the admission of the candidate will be cancelled.
Candidates must note that the benefit of reservation will be given to them subject to
verification of documents. If it is discovered at any stage that a candidate has used a
false/fake/incorrect document, or has furnished false, incorrect, or incomplete information,
in order to avail the benefit of reservation, then such a candidate shall be excluded from all
admission processes. In case such a candidate has already been given admission, the
admission shall stand cancelled.
Note:
1.
The minimum degree of disability should be 40% (Benchmark Disability) in order
to be eligible for availing reservation for persons with specified disability.
2.
The extent of “specified disability” in a person shall be assessed in accordance with
the “Guidelines for the purpose of assessing the extent of specified disability in a
person included under the Rights of Persons with Disabilities Act, 2016 (49 of
2016)” notified in the Gazette of India by the Ministry of Social Justice and
Empowerment [Department of Empowerment of Persons with Disabilities
(Divyangjan)] on 4 January 2018.
3.
No change in the category will be entertained after the last date specified by NTA for
JEE (Main) - 2024 Registration.
5.2 (b) Guidelines for conducting written examination for persons with specified disabilities
covered under the definition of Section 2(s) of the RPwD Act, 2016 but not covered
under the definition of Section 2(r) of the said Act, i.e. persons having less than 40%
disability and having difficulty in writing
The NTA does not guarantee any change in the category or sub-category (PwD status) after the
submission of the Online Application Form, and in any case, no change will be entertained by NTA
after the declaration of NTA Score for JEE (Main) - 2024. The category/sub-category (PwD status)
entered in the JEE (Main) Database by the candidate will be used for the JEE (Advanced).
Therefore, the candidates are advised to fill in the category/sub-category column very carefully.

25
I. These guidelines may be called Guidelines for conducting written examinations for
persons with specified disabilities covered under the definition of Section 2(s) of the RPwD
Act, 2016 but not covered under the definition of Section 2(r) of the said Act, i.e. persons
having less than 40% disability and having difficulty in writing.
II. The facility of scribe and/or compensatory time shall be granted solely to those having
difficulty in writing subject to the production of a certificate to the effect that the person
concerned has limitation to write and that scribe is essential to write the examination on
his/her behalf from the competent medical authority of a Government healthcare institution
as per proforma at Annexure-IV.
III. The medical authority for the purpose of certification as mentioned in point (II) above
should be a multi-member authority comprising the following:-
i. Chief Medical officer/Civil Surgeon/Chief District Medical Officer.....Chairperson
ii. Orthopaedic/PMR specialist
iii. Neurologist, if available*
iv. Clinical Psychologist/Rehabilitation Psychologist/ Psychiatrist/Special Educator
v. Occupational therapist, if available*
vi. Any other expert based on the condition of the candidate may be nominated by the
Chairperson. (* the Chief Medical Officer/Civil Surgeon/Chief District Medical Officer
may make full efforts for inclusion of neurologists, occupational therapists from the
nearest District or the Medical College/Institute if the same is not available in the
District)"
IV. Compensatory time of not less than 20 minutes per hour of the examination should be
allowed for persons who are eligible for getting a scribe. In case the duration of the
examination is less than an hour, then the duration of the compensatory time should be
allowed on a pro-rata basis. Compensatory time should not be less than 5 minutes and
should be in the multiple of 5.
Physical Disability certificate (Unique Disability Identification (UDID) issued by the notified
medical authority needs to be uploaded in the Online Application Form of JEE (Main) – 2024.
5.3 In the case of the Institutes run/aided/recognized by State Governments, the reservation policy
of the respective State Governments shall be applicable.
Notes:
1. The benefit of reservation for admission to NITs/IIITs and CFTIs shall be given only to
those classes/castes/tribes which are in the respective Central List published by the
Government of India from time to time.
2. The benefit of reservation will be given only to those castes and tribes that are mentioned
in the respective central list of corresponding states published by the Government of India
(websites:
http://socialjustice.nic.in
and
https://ncst.nic.in
).

26
CHAPTER 6
CHOICE OF CITIES
The Cities where the JEE (Main) – 2024 Session 1 will be conducted are given in Appendix I.
While applying,
candidates have to
select any four cities of their choice
.
Efforts will be made
to
allot the
city of examination to
the
candidates in order of choice opted by them in their online
Application Form.
However, due to administrative/logistic reasons,
a different city can be
allotted.
In case, there are very few candidates from a City, the NTA reserves the right to merge one, two,
or more cities.
The decision of the NTA regarding the allotment of the City/Centre shall be final. No further
correspondence or request shall be entertained in such a case.
While filling up the Online Application Form, candidates should select four Convenient Cities
from Appendix I as their Choice of Cities for the exam Centre in order of their priority.
Choice of Centre Cities will be limited to the State of Permanent Address or State of Present
Address only.
For convenience, the candidates may select their own city or neighbouring cities in their State of
Residence only and not for far away cities in other States. NTA will not be responsible for any
inconvenience due to the incorrect selection of cities by the candidates.
In case, the number of candidates opting for a city as a Centre with a regional language is less
than a certain minimum, candidates may be allotted a Centre in another city irrespective of the
city opted by the candidates for appearing in the examination.
Though every effort will be made to allot a Centre in one of the cities selected by the
candidate, the NTA reserves the right to allot a Centre in a city other than the candidate’s
choice.
Allotment of Centres will be done mostly in one of the preferred cities of the candidates and
there will be no human intervention.
Note: All efforts will be made by NTA to consider the comfort and convenience of candidates, including
PwD Candidates while allotting Centres.

27
CHAPTER 7
PARTICULARS TO BE FILLED IN THE ONLINE APPLICATION FORM
The Columns to be filled in the Online Application Form of JEE (Main) – 2024 are as follows. All
labels for the particulars will be bilingual. All candidates shall fill up the information as given below:
S. No. Page Section Fields Remarks
1.
Registration Form
Login and Identification
Log in and identify through
Digilocker
Candidates need to select any one
2.
Create a Digilocker Account
login and identify through
Digilocker
3.
Log in and identify through
ABC ID
4.
Create ABC ID and login and
identify through ABC ID
5.
Login using Indian Passport
Number
6.
Login using PAN Card
Number
7.
Login using the Aadhaar
Enrolment Number
8.
Login using Non-Indian
Passport
9.
Authentication at the Exam
Centre
Authenticate at the Exam
Centre through Aadhaar
Candidates need to select any one
10.
Authenticate at the Exam
Centre through a valid
Government ID with a
Photograph
11.
Personal Details
Candidate Name
12.
Father / Guardian Name
13.
Mother / Guardian Name
14.
Candidate's Date of Birth
15.
Gender
16.
Identity Type
17.
Identification Number
18.
Present Address
Address
19.
Locality Optional
20.
City/Town/Village
21.
Country From Drop Down
22.
State From Drop Down
23.
District From Drop Down
24.
Pincode
25.
Email Address
26.
Confirm Email Address
27.
Parent / Guardian Email
Address
28.
Mobile Number
29.
Confirm Mobile Number
30.
Parent /Guardian Mobile
Number
31.
Emergency Contact no
32.
Permanent Address Address

28
S. No. Page Section Fields Remarks
33.
Locality Optional
34.
City/Town/Village
35.
Country From Drop Down
36.
State From Drop Down
37.
District From Drop Down
38.
Pincode
39.
Choose Password
Password
40.
Confirm Password
41.
Security Question
42.
Security Answer
43.
Security Pin Security Pin to be entered as displayed on the screen
44.
Application Form
Personal Details
Candidate Name
45.
Candidate's Date of Birth
46.
Gender
47.
Father Name
48.
Mother Name
49.
Nationality
50.
State/ UT from where 12th/
Equivalent Passed/
Appearing
51.
Category
52.
If you are a PwD candidate,
do you have a benchmark
disability (40% or more)?
53.
Type of Disability only visible if PwD is yes
54.
Do you need a scribe (as per
the criteria given in the
Information Bulletin)
only visible if PwD is yes
55.
Are you Diabetic?
56.
Place of residence (Rural or
Urban)
57.
Annual Family Income From Drop Down
58.
Mode of Preparation From Drop Down
59.
Medium of Schooling in
Qualifying Examination
60.
Exam and Center Details
Applying for
All permissible combinations of Paper 1
and Paper 2
(From Drop Down )
61.
Session(s) of Exam Applying
For
62.
Question Paper Medium From Drop Down
63.
Exam State - City Choice 1 From Drop Down
64.
Exam State - City Choice 2 From Drop Down
65.
Exam State - City Choice 3 From Drop Down
66.
Exam State - City Choice 4 From Drop Down
67.
Pass Status

29
S. No. Page Section Fields Remarks
68.
Qualification Details (for class
10th or equivalent)
Year of Passing/Appearing
69.
Qualifying Exam 10th or Equivalent (pre-filled)
70.
Place of schooling (Rural or
Urban)
71.
Type of School/College From Drop Down
72.
Qualifying Examination
State of School/College
From Drop Down
73.
Qualifying Examination
District of School/College
From Drop Down
74.
School Board From Drop Down
75.
Result Mode From Drop Down
76.
Maximum Grade Point
visible if the result mode selected is
CGPA
77.
CGPA Obtained
78.
CGPA
79.
Total Marks
visible if the result mode selected is the
percentage
80.
Obtained Marks
81.
Marks(%)
82.
Roll Number
83.
School/College Name
Address
School name should be as per
mentioned in the Class 10 Marksheet
84.
School/College Pincode
85.
Qualification Details (for class
12th or equivalent)
Pass Status
86.
Year of Passing/Appearing
87.
Qualifying Exam 12th or equivalent (pre-filled)
88.
Place of schooling (Rural or
Urban)
89.
Type of School/College From Drop Down
90.
Qualifying Examination
State
From Drop Down
91.
Qualifying Examination
District
From Drop Down
92.
School Board From Drop Down
93.
Result Mode From Drop Down
94.
Maximum Grade Point
visible if the result mode selected is
CGPA
95.
CGPA Obtained
96.
CGPA
97.
Total Marks
visible if the result mode selected is the
percentage
98.
Obtained Marks
99.
Marks(%)
100.
Roll Number
101.
School/College Name
address
School name should be as per
mentioned in the Class 12 Marksheet
102.
School/College Pincode
103.
Additional Details
Are you a Twin
104.
Name of other Brother/Sister
as per Class X Certificate
only visible if 'Are you a Twin' is yes

30
S. No. Page Section Fields Remarks
105.
Roll Number of Class 10th or
its equivalent of the Twin
only visible if 'Are you a Twin' is yes
106.
Parents Income Details
Father Qualification Mandatory
107.
Father Occupation Mandatory
108.
Mother Qualification Mandatory
109.
Mother Occupation Mandatory
110.
Guardian Qualification Mandatory
111.
Guardian Occupation Mandatory
112.
Document upload Upload images
Candidate’s photograph
The size of the scanned photograph
should be between 10 kb to 200 kb
113.
Candidate’s signature
The size of the scanned signature should
be between 4 kb to 30 kb
114.
PwD certificate (if
applicable)
The size of the scanned copy of the
PwD certificate should be in pdf
between 50 kb to 300 kb

31
CHAPTER 8
ADMIT CARD
e-Admit Card
The e-Admit Card will be issued provisionally to the candidates through the NTA website:
https://jeemain.nta.ac.in/, subject to the fulfillment of the eligibility conditions and receipt of the
prescribed application fee by NTA.
The candidate has to download the Admit Card from the NTA website. The candidate will appear
in the examination at the given Centre on the date and shift/timing as indicated in his/her e-Admit
Card.
No candidate will be allowed to appear at the examination Centre, on the date and shift/time
other than that allotted to him/her in his/her Admit Card.
In case a candidate is unable to download his/her Admit Card from the website, he/she should
approach the NTA Help Line Number: 011-40759000 between 10.00 A.M. to 05.00 P.M.
The candidates are advised to read the instructions on the Admit Card carefully and strictly
follow them during the examination.
In case of any discrepancy in the particulars of the candidate or his/her photograph and signature
shown in the e-Admit Card and Confirmation Page, the candidate may immediately approach the
NTA Help Line from 10:00 A.M. to 05:00 P.M. In such a case, the candidate would appear in
the examination with the already downloaded Admit Card. However, NTA will take necessary
action to make corrections in the record later.
Note:
a. The Candidates may please note that Admit Cards will not be sent by post.
b. In no case, the duplicate Admit Card for Joint Entrance Examination (Main) - 2024
would be issued at the Examination Centres.
c. The candidates must not mutilate the Admit Card or change any entry made therein.
d. Candidates are advised to preserve their Admit Cards in good condition for future
reference.
e. No Admit Card shall be issued to the candidates whose applications are found to be
incomplete for any reasons (including indistinct/ doubtful photographs/unsigned
Applications) or who do not fulfill the eligibility criteria for the examination.
f. Issue of Admit Cards, however, shall not necessarily mean acceptance of eligibility
which shall be further scrutinized at subsequent stages of the admission process.

32
CHAPTER 9
SCHEDULE OF EXAMINATION
Schedule of Examination
Dates of Examination
Session 1: between 24 January and 01 February, 2024
Session 2: between 01 April, 2024 and 15 April, 2024
(Exact date, shift, and city of examination for Paper 1: B.E./B. Tech
and Paper 2A: B. Arch and Paper 2B: B. Planning will be made
available to the Candidates on their Admit Cards)
Mode of Examination
Paper 1:
B.E./B.Tech
“Computer Based Test (CBT)” mode only
Paper 2A:
B.Arch.
Part-I: Mathematics and
Part-II: Aptitude Test in Computer Based Test
(CBT) mode only
Part-III: Drawing Test in “Pen and Paper”
(offline) mode, to be attempted on the Drawing
sheet
of A4 size.
Paper 2B:
B. Planning
Part-I: Mathematics
Part-II: Aptitude Test, and Part-III: Planning
Based MCQs in “Computer Based Test (CBT)”
mode only
Timing of
Examination
Duration of
Examination
First Shift
Second Shift
For 3 Hours
09.00 A.M. to 12.00
Noon (IST)
03.00 P.M. to 06.00 P.M.
(IST)
For 3 Hours 30
Minutes
09.00 A.M. to 12.30
P.M. (IST)
03.00 P.M. to 06.30 P.M.
(IST)
Entry in the
Examination
Centre/Hall/Room,
frisking, biometric
registration/ record of
manual attendance by
Invigilator, document
verification/cross-
checking of Admit
Card, signature, and
photo match to be
completed by
Invigilator, etc.
07.00 A.M.. to 08.30 A.M.
01.00 P.M. to 02.30
P.M.
Instructions by the
Invigilator(s)
08.30 A.M. to 08.50 A.M.
02.30 P.M. to 02.50
P.M.
Candidates log in to
read instructions
08.50 A.M. 02.50 P.M.
Test Commences 09.00 A.M. 03.00 P.M.
Note: The duration of examination for both B.Arch. and B.Planning is 3
hrs.

33
CHAPTER 10
IMPORTANT INSTRUCTIONS FOR THE CANDIDATES
1. Candidates are advised to report at the Examination Center well on time i.e. 02 hours before
the commencement of the examination.
2. Candidates should take their seats immediately after the opening of the Examination Hall. If
the candidates do not report on time due to any reason i.e. traffic jam, train/bus delay, etc,
they are likely to miss some of the important instructions to be announced in the Examination
Rooms/Halls. The NTA shall not be responsible for any delay.
3. The candidate must show, on-demand, the Admit Card downloaded/printed from the NTA
website for admission in the examination room/hall. The Test Centre Staff on duty is
authorized to verify the identity of candidates and may take steps to verify and confirm the
identity credentials. Candidates are requested to extend their full cooperation. Candidates who
do not possess valid Admit Cards and authorized Photo IDs shall not be permitted to take the
examination under any circumstances by the Centre Superintendent.
4. A seat indicating Roll Number will be allotted to each candidate. Candidates should find and
sit in their allocated seats only. In case a candidate ventures to change his/her seat and does
not sit on the seat allotted to him/her could face cancellation of candidature. No plea would
be entertained in this regard.
5. The candidate should ensure that the Question Paper available on the computer is as per
his/her opted subject indicated in the Admit Card. In case, the subject of the Question Paper
is other than his/her opted subject, the same may be brought to the notice of the Invigilator
concerned.
6. The candidates may approach the Centre Superintendent/Invigilator in the room for any
technical assistance, first aid emergency, or any other information during the course of the
examination.
For any queries or issues regarding the Computer Based Test, the candidates may contact on
Helpline Numbers available on the Joint Entrance Examination (Main) website.
7. In case a candidate, by furnishing false information, appears in more than one shift/date, his
candidature will be cancelled and his result will not be declared.
8. For those who are unable to appear on the scheduled date of the test for any reason, a re-test
shall not be held by the NTA under any circumstances.
Note: Candidates shall appear at their own cost at the Centre on the Date and Shift as
indicated in their Admit Card issued by the NTA. Under no circumstances the choice of
cities for the Centre and shift provided in the Admit Card shall be changed.

34
ROUGH WORK
All calculations/writing work is to be done only in the Rough Sheet provided at the Test Centre
in the Examination Room/Hall and on completion of the test, candidates must hand over the
Rough Sheets to the Invigilator on duty in the Room/Hall.
Candidates MUST bring the following documents on the day of examination at the test
centre. Candidates who will not bring these will not be allowed to sit for the examination.
a. Print copy of Admit Card along with Self Declaration (Undertaking) downloaded from
the NTA Website (a clear printout on A4 size paper) duly filled in.
b. One passport size photograph (same as uploaded on the Online Application Form) for
pasting on the specific space in the Attendance Sheet at Centre during the examination.
c. Any one of the authorized photo IDs (must be original, valid, and non-expired) – School
Identity Card/ PAN card/ Driving License/ Voter ID/ Passport/ Aadhaar Card (With
photograph)/E-Aadhaar with photograph/ Ration Card with photograph/ Class 12
Board Admit Card with photograph/ Bank Passbook with Photograph.
d. PwD certificate issued by the authorized medical officer, if claiming the relaxation under
PwD category
or
PwD Certificate regarding physical limitation in an examination to write as per
Annexures given in Information Bulletin, if claiming the relaxation under PwD
category.
e. A simple transparent Ball Point Pen.

35
CHAPTER 11
UNFAIR MEANS PRACTICES AND BREACH OF EXAMINATION RULES
11.1 Definition
Unfair Means practice is an activity that allows a candidate to gain an unfair advantage
over other candidates. It includes, but is not limited to:
a) Being in possession of any item or article which has been prohibited or can be used for
unfair practices including any stationery item, communication device, accessories, eatable
items, ornaments, or any other material or information relevant or not relevant to the
examination in the paper concerned;
b) Using someone to write an examination (impersonation) or preparing material for copying;
c) Breaching examination rules or any direction issued by NTA in connection with the JEE
(Main) - 2024 examination from time to time;
d) Assisting other candidates to engage in malpractices, giving or receiving assistance of any
kind directly or indirectly or attempting to do so;
e) Contacting or communicating or trying to do so with any person, other than the
Examination Staff, during the examination time in the Examination Centre;
f) Threatening any of the officials connected with the conduct of the examination or
threatening any of the candidates;
g) Using or attempting to use any other undesirable method or means in connection with the
examination;
h) Manipulation and fabrication of online documents viz. Admit Card, Rank Letter, Self-
Declaration, etc.;
i) Forceful entry in /exit from Examination Centre/Hall;
j) Use or attempted use of any electronic device after entering the Examination Centre;
k) Affixing/uploading of wrong/morphed photographs/signatures on the Application
Form/Admit Card/Proforma;
l) Creating obstacles in the smooth and fair conduct of the examination.
m) Not attempting the question paper for long time intervals.
n) Fidgeting and looking here and there.
o) In possession of bits of paper etc.
p) Any other malpractices declared as Unfair Means by the NTA.

36
q) Any candidate with more than one Application Number (more than one SCORE
CARDS) will be treated as UFM, even if found at a later stage, and strict action will be
taken against that Candidate.
11.2 Punishment for using unfair Means practices
During the course of, before, or after the examination if a candidate indulges in any of the above
or similar practices, he/she shall be deemed to have used unfair practices and booked under the
UNFAIR MEANS (U.F.M.) case. The candidate may be debarred for 3 years in the future
and shall also be liable for criminal action and /or any other action as deemed fit.
11.3 Cancellation of Result
The result of JEE (Main) - 2024 of the candidate(s) who indulge in Unfair means Practices will
be cancelled and will not be declared. Similarly, the result of those candidates who appear from
the Centre other than the one allotted to them or allow another candidate/person to write the
examination on his behalf will be cancelled. No plea will be entertained in this regard.

37
CHAPTER 12
DISPLAY OF ANSWER KEY FOR CHALLENGE
12.1 Display of Answer Key for Challenges
1. The NTA will display the Provisional Answer Key of the questions on the NTA website:
https://jeemain.nta.ac.in/, with a Public Notice, issued to this effect on the said website, to
provide an opportunity to the candidates to challenge the Provisional Answer Keys with a
non-refundable online payment of ₹ 200/- per question challenged as processing charges. The
provisional Answer Keys are likely to be displayed for two to three days.
2. Only paid challenges made during the stipulated time through the key challenge link will be
considered. Challenges without justification/evidence and those filed on any other medium
other than the prescribed link will not be considered.
3. The NTA decision on the challenges shall be final and no further communication will be
entertained. NTA will not inform the Candidates individually about the outcome of the
challenges made.
4. The subject experts will examine all the challenges received and then a final answer key will
be displayed and declared.
5. The result will be compiled based on the final answer key declared. No grievance with
regard to answer key(s) after the declaration of result/NTA Score of JEE (Main) - 2024
will be entertained.
12.2 Display of Question Paper attempted and Recorded Responses
The NTA will display the recorded responses and Question Papers attempted by the candidates
on the NTA website https://jeemain.nta.ac.in/ prior to the declaration of the result/NTA Score.
The recorded responses are likely to be displayed for two to three days.

38
CHAPTER 13
JEE (MAIN) NTA SCORE FOR B.E./B.TECH, B.ARCH, AND B.PLANNING
13.1 JEE (Main) NTA Score for B.E./B.Tech, B.Arch, and B.Planning
a) Evaluation of multiple-choice questions of B.E./B.Tech, B.Arch, and B.Planning will be carried
out using final
answer
keys
and the raw (actual) marks obtained by a candidate will be considered
further for computation of the result of JEE (Main) - 2024.
b) For multi-shift papers, raw (actual) marks obtained by the candidates in different shifts/sessions
will be converted to NTA Score.
c) The detailed procedure for the compilation of the NTA Score is available on the NTA
Website and also in the Appendix-V of the Information Bulletin.
13.2 Compilation and display of Result of Paper-1 (B.E./B.Tech.) of JEE (Main) - 2024:
a) Compilation and display of NTA Score for Session 1 of Paper-1 (B.E. /B.Tech.):
Since Session 1 of JEE (Main) - 2024 B.E./B.Tech will be conducted in multi-shifts, and NTA
scores will be calculated corresponding to the raw (actual) marks obtained by a candidate. The
calculated NTA scores for all the shifts will be declared on the NTA website(s). This will comprise
the NTA scores for each of the three subjects (Mathematics, Physics, and Chemistry) and the total
in B.E./B.Tech of Session 1 of Paper 1 of JEE (Main) – 2024.
b) Compilation of NTA Score for Session 2 of Paper 1 (B.E. /B.Tech.):
Similarly, Session 2 of JEE (Main) - 2024 B.E./B.Tech will be conducted in multi-shifts, and NTA
scores will be calculated corresponding to the raw (actual) marks obtained by a candidate.
The calculated NTA scores for all the shifts will comprise the two NTA scores for each of the three
subjects (Mathematics, Physics, and Chemistry) and the total in B.E./B.Tech of Session 2 of
Paper 1 of JEE (Main) – 2024.
c) Compilation and display of results including overall Merit List/Ranking of Paper 1 (B.E.
/B.Tech.):
The NTA scores for each of the candidates in Total in B.E./B.Tech for Session 1 as well as for
Session 2 of JEE (Main) - 2024 will be merged for the compilation of results and preparation of the
overall Merit List/Ranking. The best of the two NTA Scores (Total) will be considered for
further processing for those candidates who appeared in both sessions.
In case of a tie, i.e. when two or more candidates obtain equal Total NTA Score in JEE (Main)-
2024, the inter-se merit of such candidates shall be determined as per “Method of resolving ties”
given in Chapter 3 for Paper 1.[3.4 (5)]
The NTA scores and rank of B.E./B. Tech of JEE (Main) – 2024 for all candidates who appeared
in Session 1 as well as in Session 2 for JEE (Main) - 2024 will be declared on the NTA website(s).
This shall comprise the NTA scores obtained by the candidate in Session 1, NTA scores obtained
in Session 2, and NTA scores for those who appeared in both sessions (best of the two Total NTA

39
scores) along with the status of those who qualify for appearing in JEE (Advanced) – 2024 provided
and subject to other conditions of eligibility being met. The rank shall comprise the All India rank
and the All India category rank.
13.3 Compilation and display of Result of Paper 2A (B. Arch) or Paper-2B (B. Planning) of
JEE (Main)-2024:
a) Compilation and display of NTA Score for Session 1 of Paper 2A (B. Arch) or Paper 2B (B.
Planning):
Since Session 1 of JEE (Main) - 2024 Paper-2A (B. Arch) or Paper-2B (B. Planning) will be
conducted in multi-shifts, NTA scores will be calculated corresponding to the raw (actual) marks
obtained by a candidate. The calculated NTA scores for all the shifts will be declared on the NTA
website(s). This will comprise the NTA scores for each of the three parts (Mathematics, Aptitude
Test, and Drawing Test or Planning Based Test) and the total in Paper 2A (B. Arch) or Paper 2B
(B. Planning) of Session 1 of JEE (Main) – 2024.
b) Compilation of NTA Score for Session 2 of Paper 2A (B. Arch) or Paper 2B (B. Planning):
Similarly, Session 2 of JEE (Main) - 2024 Paper 2A (B. Arch) or Paper 2B (B. Planning) will be
conducted in multi-shifts, and NTA scores will be calculated corresponding to the raw (actual)
marks obtained by a candidate. The calculated NTA scores for all the shifts will comprise the NTA
scores for each of the three parts (Mathematics, Aptitude Test, and Drawing Test or Planning Based
Test) and the total in Paper 2A (B. Arch) or Paper 2B (B. Planning) of Session 2 of JEE (Main) –
2024.
c) Compilation and display of results including overall Merit List/ Ranking of Paper 2A (B.
Arch) or Paper 2B (B. Planning):
The NTA scores for each of the candidates in Total in Paper 2A (B. Arch) or Paper 2B (B.
Planning) for Session 1 as well as for Session 2 of JEE (Main)-2024 will be merged for compilation
of results and preparation of overall Merit List/Ranking. The best of the two NTA Scores (Total)
will be considered for further processing for those candidates who appeared in both sessions.
In case of a tie, i.e. when two or more candidates obtain equal Total NTA Score in JEE(Main)-
2024,inter-se merit of such candidates shall be determined as per “Method of resolving ties” given
in Chapter- 3 for Paper-2A (B. Arch) or Paper-2B (B. Planning). [3.4 (5) Part 2].
The NTA scores and rank of Paper-2A (B. Arch) or Paper-2B (B. Planning) of JEE (Main) – 2024
for all candidates who appeared in Session 1 as well as in Session 2 for JEE (Main) - 2024 will be
declared on the NTA website(s). This shall comprise the NTA scores obtained by the candidate in
Session 1, NTA scores obtained in Session 2, and NTA scores for those who appeared in both
sessions (best of the two Total NTA scores). The rank shall comprise the All India rank and All
India category rank.
A copy of the Final Scorecard of JEE (Main) – 2024 will be sent to the registered e-mail address
of the Candidates.

40
13.4. Re-Evaluation/Re-Checking of Result
There shall be no re-evaluation/re-checking of the result. No correspondence in this regard shall be
entertained.
13.5. Use of Scores of JEE (Main) – 2024 by Other Organizations
The merit list/scores/result of JEE (Main) – 2024 may be utilized by other Entities of Central/State
Governments with their eligibility criteria/norms/applicable regulations/guidelines/rules.
For the academic year 2024-25, admissions under MEA (Welfare) quota seats shall be based on the
Joint Entrance Examination (Main).
Dr. APJ Abdul Kalam Technical University (AKTU), Lucknow, and Madan Mohan Malviya
University of Science and Technology, Gorakhpur will also use the score sheet of JEE (Main) –
2024 for admission to the B.Tech./M.Tech. (Integrated)/B.Arch. Courses in its affiliated colleges
across the State.
From the Academic year 2024-25, the Government of Goa has approved that admissions to
Engineering Degree Courses in the State Engineering Colleges in Goa State will also be done
based on JEE (Main).
Note:
1 The All India Rank shall be compiled and declared after conduct of Session 2 of JEE (Main) - 2024
Examination.
2 No Score/Rank Card will be dispatched to the candidates and the candidates are advised to download
their Score/Rank Cards from the JEE (Main) website: www.nta.ac.in/, https://jeemain.nta.ac.in/ only.
3 There shall be no provision for re-valuation/re-checking of the Score. No correspondence in this regard
shall be entertained.
4 Only the All India Rank (AIR) is used for admissions through Central Seat Allocation Board (CSAB)/Joint
Seat Allocation Authority (JoSAA) to NITs/ IIITs/ CFTIs/ SFIs/ Others.
5 All participating Institutions that do not use the Centralized Seat Allocation Process will prepare their
own ranking based on the performance in JEE (Main) - 2024 and other criteria as decided by them.
6 The National Testing Agency (NTA) is an examination conducting body and not an admission giving
authority. Therefore, NTA does not collect the information regarding the total number of seats available
in the institutions, or the eligibility and reservation criteria followed by the institutions.
41
CHAPTER 14
ADMISSION
Admission to NITs, IIITs, CFTIs, SFIs, State Engineering Colleges in the Participating
States, and Other Participating Institutions.
14.1 Eligibility for Admission to NITs, IIITs, and CFTIs participating through the Central
Seat Allocation Board (CSAB):
For the candidates who qualify for admission in the NITs, IIITs and such other CFTIs whose
admissions are based on the JEE (Main) ranks, they should have secured at least 75% marks in
the Class 12 examination or be in the top 20 percentile in Class 12 examination conducted by
respective Boards. For SC / ST candidates, the qualifying marks should be 65% in the Class 12
examination
The candidate is also required to pass in each of the subjects of Class 12 / qualifying
examination.
The Admission to NITs, IIITs, and CFTIs will be based on the announced qualifying Percentile
by the individual Institutes.
For eligibility regarding details, candidates may refer to the website https://csab.ac.in/.
The eligibility criteria decided by the Council of Architecture for admission to B.Arch. course other
than NITs, IIITs, and CFTIs:
“No candidate shall be admitted to architecture course unless he has passed an examination
at the end of the 10+2 scheme of examination with at least 50 percent. aggregate marks in
Physics, Chemistry, and Mathematics and also at least 50 percent marks in aggregate of the
10+2 level examination or passed the 10+3 Diploma Examination with Mathematics as a
compulsory subject with at least 50 percent. marks in aggregate.”
For B. Planning, the candidates should have passed the qualifying examination with 50 % marks
in Mathematics and 50% marks in aggregate of the qualifying examination.
Subject combinations required in the qualifying examination for admission to B.E./B.Tech, B. Arch,
and B. Planning Courses in NITs, IIITs, and other CFTIs shall be as under.
Course
Required Criteria based on Class 12/ Equivalent qualifying Examination
B.E/B.Tech.
Passed qualifying examination with Physics and Mathematics as compulsory
subjects along with one of the Chemistry/Biotechnology/Biology/ Technical
Vocational subjects.

42
The letters / e-mails / grievances / queries / RTI applications / Court cases pertaining to
admission related matters/procedures will not be entertained by NTA. The same may be
addressed to the concerned Counseling/Authorities/Admitting Institutions.
The Admission Policy, as announced by the Competent Authority of the admitting institutes
shall be followed at the time of admission. The Candidates are advised to satisfy themselves
about their Eligibility for Admission from the respective websites of admitting authorities.
14.2 Eligibility for Admission to Other Institutions
The above-mentioned policy could also be adopted by other Technical Institutions participating in
counseling through JoSAA/CSAB. In case a State opts to admit students to the engineering
Colleges affiliated with State Universities, the State may prepare a separate rank list based on
criteria decided by them.
For all admission-related procedures/queries, the candidates are advised to refer to the website of
JoSAA, Central Seat Allocation Board (CSAB), or the concerned State Government/Institute after
the declaration of result/ranks of JEE (Main) - 2024.
14.3 Seat Allocation Process and Admission Procedure
Candidates shall be offered admission based on their choices and All India Ranks of JEE (Main) -
2024 through a Seat Allocation Process to be announced later by CSAB/JoSAA. The
authentication/verification of relevant documents in support of identification, date of birth,
qualifying examination, State of eligibility, category, and disability (if any) of the qualifying
candidates would be done at the time of the Seat Allocation/ admission process. On failing to
produce any of the authentic documents, the candidate may not be considered for admission.
Gen-EWS, SC, ST, OBC, and PwD candidates will be required to produce the original certificate
as per prescribed formats issued by the competent authority at the time of the Seat
Allocation/admission process failing which they will not be considered for admission.
B.Arch.
No candidate shall be admitted to an architecture course unless he has passed
an examination at the end of the 10+2 scheme of examination with at least 50
percent. aggregate marks in Physics, Chemistry, and Mathematics and also at
least 50 percent marks in aggregate of the 10+2 level examination or passed
the 10+3 Diploma Examination with Mathematics as a compulsory subject
with at least 50 percent. marks in aggregate.
B.Planning Passed qualifying examination with Mathematics

43
14.4 Eligibility Criteria for Indian Nationals Appearing in JEE (Advanced) - 2024
(Please refer to the JEE (Advanced) website https://jeeadv.ac.in/ for the latest information)
All the candidates must simultaneously fulfill each one of the following to appear for JEE
(Advanced) - 2024.
Performance in JEE (Main) - 2024:
Candidates should be among the top 2,50,000 successful candidates (including all categories) in
B.E./B.Tech. Paper of JEE (Main) - 2024.
Number of Attempts:
A candidate can attempt JEE (Advanced) a maximum of two times in two consecutive years.

44
CHAPTER 15
NATIONAL TEST ABHYAS
15.1 National Test Abhyas
The National Testing Agency (NTA), India’s premier autonomous testing organization for
conducting entrance examinations for admission in higher education institutions, launched a
mobile app called the “National Test Abhyas” - to enable candidates to take mock tests for
various competitive exams such as JEE (Main), NEET (UG), UGC-NET, and other exams under
the NTA’s purview. The app had been launched to facilitate candidates’ access to high-quality
mock tests in the safety and comfort of their homes since NTA’s Test-Practice Centers (TPCs)
were closed during the ongoing COVID-19 pandemic.
With this facility for the candidates, India has taken the lead in restoring a semblance of
normalcy in one more crucial area – education – even as we tackle these unprecedented times
that have led to significant changes in life around the world.
Candidates across the country can use the App to access high-quality tests, free of cost, in a bid
to be fully prepared for the upcoming JEE (Main), NEET (UG), and other competitive exams.
The tests can be easily downloaded and taken offline, thus accommodating candidates with low
bandwidth internet connections.
The app works on Android-based and iOS smartphones and tablets and can be downloaded from
Google Play Store or App Store. Once candidates download the app, they simply need to sign-
up or register with some basic details, create a free account, and then start accessing mock tests
free of cost for their selected examination(s).
In addition to that NTA has developed an extensive support system at
http://nta.ac.in/abhyas/help.
The National Test Abhyas is available on the website: https://www.nta.ac.in/Abhyas and Play
Store: https://play.google.com/store/apps/details?id=com.abhyas.nta.com

45
CHAPTER 16
MISCELLANEOUS
16.1 Query Redressal System (QRS)
National Testing Agency (NTA) has established a Query Redressal System (QRS), an online
web-enabled system developed by NTA. QRS is the platform based on web technology that
primarily aims to enable the submission of queries/grievances by the Registered Candidate(s) of
the JEE (Main)-2024 Examination with (a 24x7) facility for speedy and favorable redressal of
the queries/grievances. A Unique Registration Number will be generated for tracking the status
of the queries/grievances.
The Registered Candidate(s) are advised to use the online facility for a speedy response.
16.2 Correspondence with NTA
All the correspondence should preferably be addressed by e-mail. The email query shall be
addressed only if it is not anonymous and contains the name, postal address, and contact number
of the sender. An email containing vague or general queries and other queries as contained in
the Information Bulletin shall not be entertained. Queries shall not be entertained from a person
claiming to be representatives, associates, or officiates of the applicant candidate. The following
information shall not be revealed by phone or email:
a. Internal documentation/status.
b. The internal decision-making process of NTA. Any claim/counterclaim thereof.
c. Dates and venue of internal meetings or name of the staff/officers dealing with it.
d. Any information which cannot be revealed in the opinion of NTA.
16.3 Weeding Out Rules
The record of Joint Entrance Examination JEE (Main) - 2024 will be preserved up to 90 days
from the date of declaration of result.
16.4 Legal Jurisdiction
All disputes pertaining to the conduct of the JEE (Main) – 2024 Examination including Results
shall fall within the jurisdiction of Delhi/New Delhi only. Further, any grievance/representation
arising out of the Result shall be entertained only when raised within 30 days from the declaration
of the result. Further, the copy of the Legal Notice or Writ Petition/Petition/Appeal/Reply may
be served to NTA only through: [email protected]n
16.5 RTI
Information uploaded on the website shall not be provided to the candidate or any other person
under R.T.I. Act, 2005. The information uploaded on the website shall remain for a specific
period only. Therefore, the candidates are advised to download the uploaded information and
keep it with them for the future. In due course of examination or in the midway of process,
neither any application under the Right to Information Act, 2005 shall be entertained nor
information will be provided.

46
Annexures

47
Annexure-I
Form-PwD (IV)
Disability Certificate
Certificate for persons with a disability under the RPwD Act, 2016 having less than 40% disability
This is to certify that, we have examined Mr/Ms/Mrs …………. (name of the candidate), S/o /D/o
……………………, a resident of ………………(Vill/PO/PS/District/State), aged …………….. yrs, a
person with ……………………. (nature of disability/condition), and to state that he/she has a limitation
which hampers his/her writing capability owing to his/her above condition. He/she requires the support of
a scribe for writing the examination.
2. The above candidate uses aids and an assistive devices such as prosthetics & orthotics, and hearing aids
(name to be specified) which is /are essential for the candidate to appear at the examination with the
assistance of a scribe.
3. This certificate is issued only for the purpose of appearing in written examinations conducted by
recruitment agencies as well as academic institutions and is valid upto __________ (it is valid for a
maximum period of six months or less as may be certified by the medical authority)
Signature of medical authority
Name of Government Hospital/Health Care Centre with Seal
Place:
Date:
(Signature & Name) (Signature & Name) Signature &
Name)
(Signature &
Name)
(Signature &
Name)
Orthopedic / PMR
specialist
Clinical Psychologist/
Rehabilitation
Psychologist/Psychiatrist
/ Special Educator
Neurologist
(if available)
Occupational
therapist (if
available)
Other
Expert, as
nominated
by the
Chairperson
(if any)
(Signature & Name)
Chief Medical Officer/Civil Surgeon/Chief District Medical Officer………Chairperson
Recent PP size
attested photograph
(showing face only)
of the person with
disability

48
Annexure-II
Letter of Undertaking for Using Own Scribe
I __________________________, a candidate with_______________________(name of the disability)
appearing for the ___________________________(name of the examination) bearing Application No.
__________________at_____________________ (name of the centre) in the District __________,
______________________________ (name of the State). My qualification is ___________________________.
I do hereby state that_________________________________(name of the scribe) will provide the service of
scribe/reader/lab assistant for the undersigned for taking the aforesaid examination.
I do hereby undertake that his qualification is _________________________________. In case, subsequently,
it is found that his qualification is not as declared by the undersigned and is beyond my qualification, I shall
forfeit my right to the admission and claims relating thereto.
(Signature of the candidate with disability)
Place:
Date:
(Self-Attested Photograph)
Name of Scribe
ID of the Scribe
ID Number
Photograph of Scribe

49
Appendix - I
List of Examination Cities for JEE (Main) - 2024
JEE (Main) - 2024 will be conducted in the following Cities, provided there are sufficient numbers of
candidates in the City:
State-wise/UT-wise list of Cities in India:
State / Country City City Code
Andaman and Nicobar Island Port Blair AN01
Andhra Pradesh Amalapuram AP35
Andhra Pradesh Anantapur AP01
Andhra Pradesh Bobbili AP36
Andhra Pradesh Chirala AP04
Andhra Pradesh Chittoor AP05
Andhra Pradesh Eluru AP06
Andhra Pradesh Gooty AP37
Andhra Pradesh Gudlavalleru AP38
Andhra Pradesh Guntur AP07
Andhra Pradesh Kadapa AP08
Andhra Pradesh Kakinada AP09
Andhra Pradesh Kurnool AP10
Andhra Pradesh Madanapalle AP39
Andhra Pradesh Markapur AP40
Andhra Pradesh Nandyal AP29
Andhra Pradesh Nellore AP11
Andhra Pradesh Ongole AP12
Andhra Pradesh Papum Pare AL02
Andhra Pradesh Proddatur AP21
Andhra Pradesh Puttaparthi AP41
Andhra Pradesh Puttur AP AP42
Andhra Pradesh Rajahmundry AP13
Andhra Pradesh Srikakulam AP14
Andhra Pradesh Tadpatri AP43
Andhra Pradesh Tirupathi AP16
Andhra Pradesh Tiruvuru AP44
Andhra Pradesh Vijayawada AP17
Andhra Pradesh Visakhapatnam AP18
Andhra Pradesh Vizianagaram AP19
Arunachal Pradesh Itanagar/Naharlagun AL01
Assam Dibrugarh AM06
Assam Guwahati AM02
Assam Jorhat AM03
Assam Lakhimpur AS AM07
Assam Silchar (Assam) AM04

50
State / Country City City Code
Assam Tezpur AM05
Bihar Arrah BR09
Bihar Aurangabad(Bihar) BR01
Bihar Bhagalpur BR02
Bihar Darbhanga BR04
Bihar Gaya BR05
Bihar Muzaffarpur BR06
Bihar Patna BR07
Bihar Purnea BR08
Bihar Rohtas BR41
Bihar Samastipur BR12
Chhattisgarh Bhilai Nagar/Durg CG01
Chhattisgarh Bilaspur(Chhattisgarh) CG02
Chhattisgarh Raipur CG03
Chhattisgarh Jagdalpur CG04
Chhattisgarh Ambikapur CG05
Dadra and Nagar Haveli Silvassa DN01
Delhi/New Delhi Delhi/New Delhi DL01
Goa Panaji GO03
Gujarat Ahmedabad GJ01
Gujarat Gandhinagar GJ32
Gujarat Anand GJ02
Gujarat Himatnagar GJ14
Gujarat Jamnagar GJ06
Gujarat Mehsana GJ31
Gujarat Rajkot GJ10
Gujarat Surat GJ11
Gujarat Vadodara GJ12
Gujarat Valsad/Vapi GJ13
Haryana Faridabad HR03
Haryana Ambala HR01
Haryana Hisar HR10
Himachal Pradesh Bilaspur (Himachal Pradesh) HP15
Himachal Pradesh Hamirpur (Himachal Pradesh) HP03
Himachal Pradesh Kangra HP16
Himachal Pradesh Kullu HP10
Himachal Pradesh Mandi HP08
Himachal Pradesh Shimla HP06
Himachal Pradesh Solan HP17
Himachal Pradesh Una HP09
Jammu and Kashmir Anantnag JK05
Jammu and Kashmir Baramulla JK01
Jammu and Kashmir Budgam JK11

51
State / Country City City Code
Jammu and Kashmir Jammu JK02
Jammu and Kashmir Pulwama JK06
Jammu and Kashmir Samba JK12
Jammu and Kashmir Srinagar(J & K) JK04
Jharkhand Bokaro JH01
Jharkhand Dhanbad JH02
Jharkhand Hazaribagh JH05
Jharkhand Jamshedpur JH03
Jharkhand Ranchi JH04
Karnataka Bagalkot KK19
Karnataka Ballari KK03
Karnataka Belagavi(Belgaum) KK02
Karnataka Bengaluru KK04
Karnataka Bengaluru- Urban KK20
Karnataka Chikaballapur KK22
Karnataka Chikmagalur KK23
Karnataka Davangere KK06
Karnataka Dharwad KK10
Karnataka Gulbarga/Kalaburgi KK08
Karnataka Hassan KK09
Karnataka Hubli KK27
Karnataka Kolar KK30
Karnataka Mandya KK18
Karnataka Mangaluru(Mangalore) KK12
Karnataka Mysuru(Mysore) KK14
Karnataka Shivamoga(Shimoga) KK15
Karnataka Tumakuru KK16
Karnataka Udupi/Manipal KK17
Kerala Alappuzha/Chengannur KL01
Kerala Ernakulam/Moovattupuzha KL04
Kerala Angamaly KL20
Kerala Idukki KL05
Kerala Kannur KL07
Kerala Kasaragod KL08
Kerala Kollam KL09
Kerala Kottayam KL11
Kerala Kozhikode/Calicut KL12
Kerala Malappuram KL13
Kerala Palakkad KL15
Kerala Pathanamthitta KL16
Kerala Thiruvananthapuram KL17
Kerala Thrissur KL18
Kerala Wayanad KL19

52
State / Country City City Code
Ladakh Kargil LL02
Ladakh Leh LL01
Lakshadweep Kavaratti LD01
Madhya Pradesh Balaghat MP01
Madhya Pradesh Bhopal MP03
Madhya Pradesh Gwalior MP06
Madhya Pradesh Indore MP07
Madhya Pradesh Jabalpur MP08
Madhya Pradesh Khandwa MP29
Madhya Pradesh Sagar MP12
Madhya Pradesh Satna MP13
Madhya Pradesh Ujjain MP15
Maharashtra Ahmednagar MR01
Maharashtra Akola MR02
Maharashtra Amravati MR03
Maharashtra Aurangabad (Maharashtra) MR04
Maharashtra Beed MR30
Maharashtra Bhandara MR31
Maharashtra Buldhana MR32
Maharashtra Chandrapur MR09
Maharashtra Dhule MR10
Maharashtra Gondia MR35
Maharashtra Jalgaon MR13
Maharashtra Kolhapur MR14
Maharashtra Latur MR15
Maharashtra Mumbai/Navi Mumbai MR16
Maharashtra Nagpur MR17
Maharashtra Nanded MR18
Maharashtra Nandurbar MR36
Maharashtra Nashik MR19
Maharashtra Parbhani MR38
Maharashtra Pune MR22
Maharashtra Raigad MR23
Maharashtra Ratnagiri MR24
Maharashtra Sangli MR25
Maharashtra Satara MR26
Maharashtra Sindhudurg MR39
Maharashtra Solapur MR27
Maharashtra Thane MR28
Maharashtra Wardha MR29
Maharashtra Yavatmal MR34
Manipur Imphal MN01
Meghalaya Shillong MG01

53
State / Country City City Code
Meghalaya Tura MG02
Mizoram Aizawl MZ01
Nagaland Dimapur NL01
Nagaland Kohima NL02
Odisha Angul OR10
Odisha Balangir OR20
Odisha Balasore (Baleswar) OR02
Odisha Baragarh OR21
Odisha Baripada/Mayurbanj OR12
Odisha Berhampur / Ganjam OR03
Odisha Bhadrak OR11
Odisha Bhawanipatna OR30
Odisha Bhubaneswar OR04
Odisha Cuttack OR05
Odisha Dhenkanal OR06
Odisha Jagatsinghpur OR17
Odisha Jajpur OR13
Odisha Jeypore (Odisha) OR19
Odisha Jharsuguda OR22
Odisha Kendrapara OR14
Odisha Nuapada OR31
Odisha Paralakhemundi (Gajapati) OR24
Odisha Phulbani (Kandhamal) OR25
Odisha Puri OR16
Odisha Rayagada OR26
Odisha Rourkela OR08
Odisha Sambalpur OR09
Puducherry Puducherry PO01
Punjab Amritsar PB01
Punjab Bhatinda PB02
Punjab Chandigarh CH01
Punjab Jalandhar PB04
Punjab Ludhiana PB05
Punjab Moga PB20
Punjab Pathankot PB07
Punjab Patiala/Fatehgarh Sahib PB08
Punjab Sahibzada Ajit Singh Nagar PB12
Rajasthan Ajmer RJ01
Rajasthan Alwar RJ02
Rajasthan Bhilwara RJ12
Rajasthan Bikaner RJ05
Rajasthan Dausa RJ17
Rajasthan Hanumangarh RJ23

54
State / Country City City Code
Rajasthan Jaipur RJ06
Rajasthan Jodhpur RJ07
Rajasthan Kota RJ08
Rajasthan Sikar RJ09
Rajasthan Sriganganagar RJ10
Rajasthan Udaipur RJ11
Sikkim Gangtok SM01
Tamil Nadu Chennai TN01
Tamil Nadu Coimbatore TN02
Tamil Nadu Cuddalore TN03
Tamil Nadu Coonoor TN36
Tamil Nadu Dharmapuri TN26
Tamil Nadu Dindigul TN27
Tamil Nadu Erode TN28
Tamil Nadu Kanchipuram TN05
Tamil Nadu Kanyakumari/Nagercoil TN06
Tamil Nadu Karur TN29
Tamil Nadu Krishnagiri TN21
Tamil Nadu Kallakurichi TN37
Tamil Nadu Madurai TN08
Tamil Nadu Nagapattinam TN30
Tamil Nadu Namakkal TN10
Tamil Nadu Pudukkottai TN31
Tamil Nadu Ramanathapuram TN32
Tamil Nadu Salem TN11
Tamil Nadu Sivaganga TN33
Tamil Nadu Thanjavur TN12
Tamil Nadu Thoothukudi TN13
Tamil Nadu Tiruchirappalli TN14
Tamil Nadu Tirunelveli TN15
Tamil Nadu Tiruppur TN22
Tamil Nadu Tiruvannamalai TN35
Tamil Nadu Vellore TN18
Tamil Nadu Viluppuram TN23
Tamil Nadu Virudhunagar TN20
Telangana Hyderabad/Secunderabad TL01
Telangana Karimnagar TL02
Telangana Khammam TL03
Telangana Kothagudem TL17
Telangana Mahbubnagar TL04
Telangana Nalgonda TL05
Telangana Nizamabad TL08
Telangana Siddipet TL11

55
State / Country City City Code
Telangana Suryapet TL09
Telangana Warangal TL07
Tripura Agartala TA01
Uttar Pradesh Ghaziabad UP07
Uttar Pradesh Meerut UP14
Uttar Pradesh Noida/Greater Noida UP09
Uttar Pradesh Agra UP01
Uttar Pradesh Aligarh UP02
Uttar Pradesh Allahabad/Prayagraj UP03
Uttar Pradesh Ambedkar Nagar UP25
Uttar Pradesh Azamgarh UP19
Uttar Pradesh Ballia UP20
Uttar Pradesh Bareilly UP04
Uttar Pradesh Bijnor UP21
Uttar Pradesh Bulandshahr UP29
Uttar Pradesh Basti UP59GJ32
Uttar Pradesh Chandauli UP41
Uttar Pradesh Faizabad UP06
Uttar Pradesh Firozabad UP22
Uttar Pradesh Ghazipur UP23
Uttar Pradesh Gorakhpur UP08
Uttar Pradesh Jhansi UP10
Uttar Pradesh Kanpur UP11
Uttar Pradesh Lucknow UP12
Uttar Pradesh Mathura UP13
Uttar Pradesh Mau UP35
Uttar Pradesh Moradabad UP15
Uttar Pradesh Muzaffarnagar UP16
Uttar Pradesh Rampur UP58
Uttar Pradesh Saharanpur UP38
Uttar Pradesh Sitapur UP17
Uttar Pradesh Sultanpur UP40
Uttar Pradesh Varanasi UP18
Uttar Pradesh Pratapgarh UP43
Uttarakhand Almora UK09
Uttarakhand Dehradun UK01
Uttarakhand Haldwani UK02
Uttarakhand Pauri Garhwal UK08
Uttarakhand Roorkee UK06
West Bengal Asansol WB01
West Bengal Bankura WB16
West Bengal Burdwan(Bardhaman) WB02
West Bengal Durgapur WB04

56
State / Country City City Code
West Bengal Hooghly WB06
West Bengal Howrah WB07
West Bengal Kalyani WB08
West Bengal Kolkata WB10
West Bengal Siliguri WB11
West Bengal Suri WB22
Bahrain Manama ZZ01
Sri Lanka Colombo ZZ02
Qatar Doha ZZ03
UAE Dubai ZZ04
Nepal Kathmandu ZZ05
Oman Muscat ZZ06
Saudi Arabia Riyadh ZZ07
UAE Sharjah ZZ08
Singapore Singapore ZZ09
Kuwait Kuwait City ZZ10
Malaysia Kuala Lumpur ZZ11
Nigeria Lagos/Abuja ZZ12
Indonesia Jakarta ZZ13
Australia Canberra ZZ14
Austria Vienna ZZ15
Brazil Brasilia ZZ16
Canada Ottawa ZZ17
Hong Kong Hong Kong ZZ19
Mauritius Port Luis ZZ20
Russia Moscow ZZ21
South Africa Cape Town ZZ22
Thailand Bangkok ZZ23
USA Washington D.C. ZZ24
Vietnam Hanoi ZZ25
Note:
1. NTA reserves the right to change any of the above-mentioned cities that may have been opted by
any candidate in his/ her online Application Form, at a later stage due to logistic reasons and may
allot another city to him/her from his/ her choices of cities, given in the online Application Form.
2. Choice of examination cities displayed to the candidates will be based on the permanent and
correspondence addresses filled during the Online Application Form of JEE (Main) – 2024.

57
Appendix –II
Procedure for Online Payment of Fees and Helpline for Payment-Related Queries
After completing Step 2 of the Online Application Form, the candidate may remit the examination fee (Step
3) in the following manner:
Check the validity of the Debit/ Credit Card and keep it ready with you while logging on to the
website for submitting the Application Form. The candidates should enter the information asked
for and make payment through a Debit/ Credit Card.
Through Net Banking, check the balance in your account and keep all credentials ready with you
while logging on to the website for making payments. The candidates should log in with his/her
credentials of net banking and make payments accordingly.
For payment through a UPI Service, check whether there is a requisite balance in the Bank Account
/ Wallet to which the concerned UPI is linked.
The prescribed Examination Fee (please see the Information at a Glance) can be paid through any
Bank/Payment Gateway in any Payment Mode [service/processing charges per transaction and GST
applicable thereon @ 18 % are to be borne by the candidate
S.
N
o
Mode of
Payment
ICICI BANK HDFC BANK CANARA BANK STATE BANK OF INDIA
1
Net
Banking
ICICI
NIL Charge
HDFC
NIL Charge
Canara Bank
NIL Charge
SBI
Rs 5.00+GST
Other
Banks
4.00 + GST
Other
Banks
4.00 + GST Other Banks 5.00 + GST
Other Banks
2
All Debit
Card
ICICI or
Other
Banks
Transact
ion upto
Rs
2000/
-
0 %
HDFC
or
Other
Banks
Transaction
upto
Rs 2000/-
0 %
Canara
Bank
or
Other
Banks
Nil
Charge
Nil Transaction upto
Rs 2000/-
Transact
ion
above
Rs
2000/
-
0.5 % +
GST
Transaction
above
Rs 2000/-
0 %
Transaction above
Rs 2000/-
3
Credit
Card
Domesti
c
0.40%of Transaction
value
Domesti
c
0.40% of Fee + GST Domestic
0.80% of
Fee + GST
Domestic
0.80% of Fee+ GST
(Minimum Rs 11/-)
Internati
onal
2.35%of Transaction
value
Internati
onal
Nil Charge International
2.35% of
Fee+ GST
International
3.50% of Fee+ GST
(Minimum Rs 11/-)
4
Unified
Payment
Interface
(UPI)
ICICI or
other
banks
Transact
ion upto
Rs
2000/
-
0 %
Nil Charge
Transaction upto
Rs 2000/-
Transact
ion
above
Rs
2000/
-
5.0 %
+ GST
Transaction above
Rs 2000/-
Note: In case, the fee payment status is not ‘OK’, or the Confirmation Page of the Online Application
is not generated after fee payment (completion of Step 3) of the candidate is advised to contact the
helpline number/email of the concerned Bank/Payment Gateway Integrator, as mentioned below:-
58
(a) If Paying through the State Bank of India (SBI):
Level
Name Email ID Contact Number
4 Through SMS UNHAPPY (add text) 8008202020
(b) If Paying through Canara Bank:
Level
Name Email ID Contact Number
9971262371
2 Complaint
Management Services
genadmnronoida@canarabank.com
01202400850/1
9971262371
4 Through SMS genadmnronoida@canarabank.com
01202400850/1
(c) If Paying through HDFC Bank:
(d) If Paying through ICICI Bank:
Level
Name Email ID
Contact Number
1 Shri Vikram Singh vikram[email protected] 9799810080
2. Amit Singh amit.singh26@hdfcbank.com 7428869770
3 Shri Ripon Bhattacharjee
ripon.bhattacharjee@hdfcbank.com
9625031697
Level
Name Email ID
Contact Number
1 Aiysha Khatun support.nta@ingenico.com 01204728426
3 Mohit Bhatia mohit.bhatia@icicibank.com 9810086455

59
Appendix-III
Computer Based Test (CBT)
The major examinations being conducted by NTA are Computer-Based Tests (CBT). A CBT requires
candidates to sit in front of a computer terminal (node) allocated to them against their Roll number and
Admit card. After logging the candidate will get detailed instructions for the examinations. At the designated
time of the start of the examination, the candidates will be able to proceed and see the questions on the
computer screen using the computer mouse. Candidates will have the option to change/modify/edit/answers
already entered at any time during the examination.
Procedure for appearing in Computer Based Test (CBT):
(Sample/mock test will be available on the NTA website: www.nta.ac.in for hands-on practice)
(a) A computer terminal (node) indicating the Roll Number will be allocated to each candidate.
Candidates should find and sit on their allocated computers only. Any candidate found to have
changed room/hall or the computer on their own other than the one allotted would lead to
cancellation of candidature and no plea in this regard would be entertained.
(b) For login, the candidate will have to enter a login ID and password. The computer terminal allotted
to the candidate will display the WELCOME login screen, the Candidate’s photograph and the
subject opted by the candidate.
Candidate Login Page
Candidate Welcome Screen
(c) After login, the candidate shall be able to see the detailed instructions for the examination.
Candidates are advised to go through the instructions carefully regarding the type of questions and
marking scheme. At the designated time of the start of the examination, the candidates will be able
to proceed and see the questions on the computer screen.
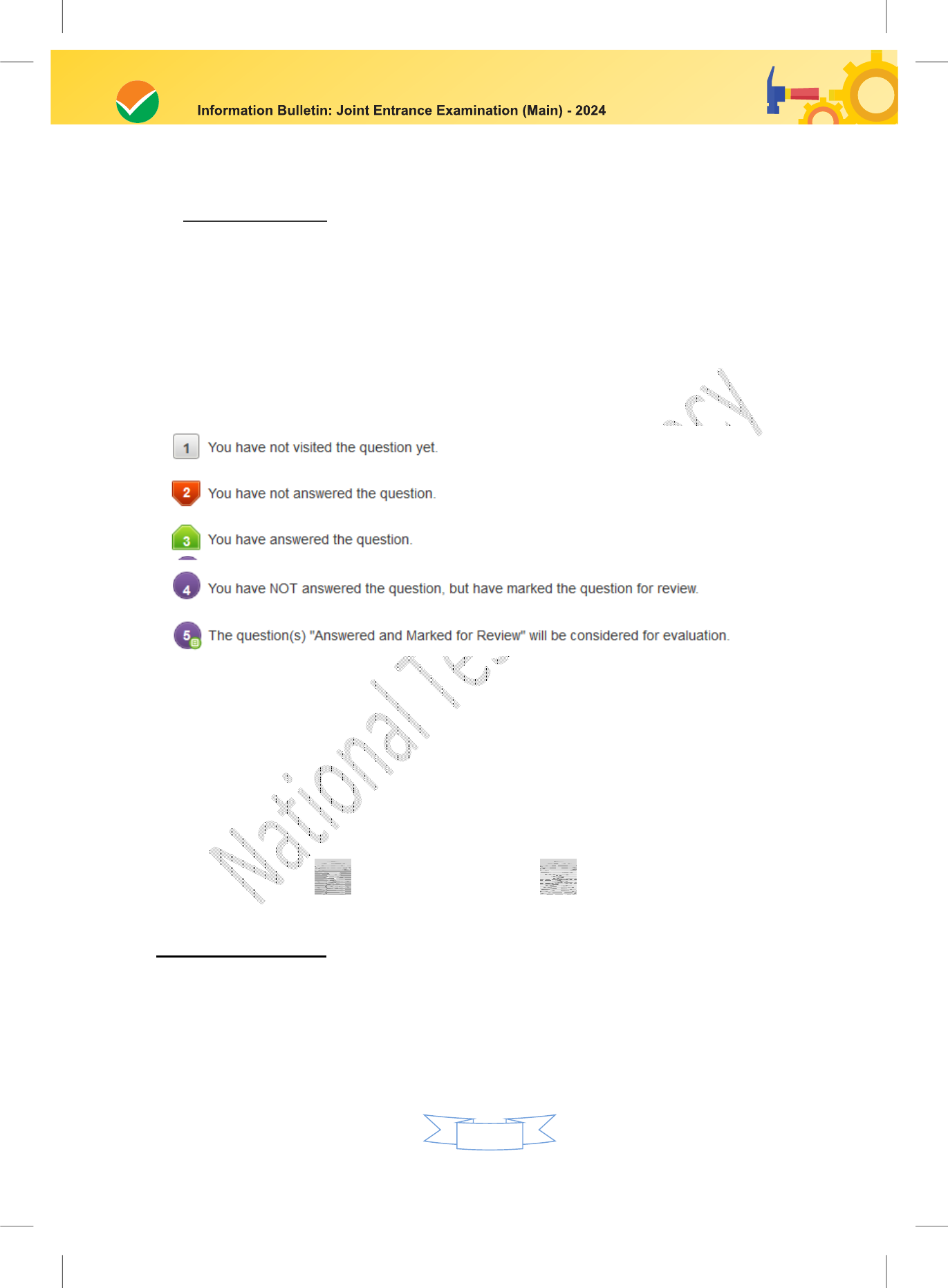
60
Please read the instructions carefully
General Instructions:
1. The total duration of the examination is 180 minutes for Paper 1 (B.E./B.Tech) and 210 minutes
for (B.Arch and B.Planning) both.
2. The clock will be set on the server. The countdown timer in the top right corner of the screen will
display the remaining time available for you to complete the examination. When the timer reaches
zero, the examination will end by itself. You will not be required to end or submit your
examination.
3. The Question Palette displayed on the right side of the screen will show the status of each question
using one of the following symbols:
4. The ‘Marked for Review’ status for a question simply indicates that you would like to look at that
question again.
5. You can click on the ">" arrow which appears to the left of the question palette to collapse the
question palette thereby maximizing the question window. To view the question palette again, you
can click on "< " which appears on the right side of the question window.
6. You can click on your "Profile" image on the top right corner of your screen to change the language
during the exam for the entire question paper. On clicking on the Profile image, you will get a
drop-down to change the question content to the desired language.
7. You can click on to navigate to the bottom and to navigate to the top of the question
area, without scrolling.
Navigating to a Question:
8. To answer a question, do the following:
a. Click on the question number in the Question Palette at the right of your screen to go to
that question directly. Note that using this option does NOT save your answer to the current
question.

61
b. Click on Save & Next to save your answer for the current question and then go to the next
question.
c. Click on Mark for Review & Next to save your answer for the current question, mark it
for review, and then go to the next question.
Answering a Question :
9. Procedure for answering a Multiple Choice Type question:
a. To select your answer, click on the button of one of the options.
b. To deselect your chosen answer, click on the button of the chosen option again or click on the
Clear Response button.
c. To change your chosen answer, click on the button of another option.
d. To save your answer, you must click on the Save & Next button
e. To mark the question for review, click on the ‘Mark for Review & Next' button.
10. To change your answer to a question that has already been answered, first select that question for
answering and then follow the procedure for answering that type of question.
Navigating through sections:
11. Sections in this question paper are displayed on the top bar of the screen. Questions in a section
can be viewed by clicking on the Section name. The section you are currently viewing is
highlighted.
12. After clicking the Save and Next button on the last question for a Section, you will automatically
be taken to the first question of the next section.
13. You can shuffle between sections and questions anytime during the examination as per your
convenience only during the time stipulated.
14. The candidate can view the corresponding section summary as part of the legend that appears in
every section above the question palette.
Instruction for Question:
15. For the correctness of the translation for a particular language, only the English version will be
considered as the final version for evaluation purposes.
The keyboard attached to the computer, if any, will be disabled during the entire duration of the
examination. Depending on the type of question, the answers to questions can either be entered by clicking
on the virtual on-screen keyboard (numeric or otherwise) using the computer mouse or by clicking the
chosen option(s) using the computer mouse. Candidates will have the option to change/modify answers
already entered at anytime during the entire duration of the examination.
In case the computer/mouse allotted to any candidate malfunctions during the test, he/she will be
immediately allotted another computer system and the time lost due to this will be adjusted in the server
so as to give the candidate the full allotted time.

62
The on-screen computer clock counter of every candidate will be set at the server. The countdown timer
on the top right side of the computer screen will display the time remaining (in minutes) available for the
candidate to complete the examination. When the timer reaches zero, the examination will end by itself.
The candidates will not be required to end or submit the examination.
The Question Palette displayed on the screen will show the status of each question using one of the
following symbols:
The question(s) “Answered and Marked for Review” status for a question indicates that the candidates
would like to have a relook at that question again. A candidate has the option of answering a question and
simultaneously “Marked for Review”, these answers will be considered for evaluation. However, if a
candidate has simply put “Marked for Review” for a question without answering it, the corresponding
question marked for review without an answer will not be considered for evaluation. It may be noted that
a candidate can return to any “Marked for Review” question at any time during the examination by
clicking on the corresponding question number icon displayed on the Question Palette of the
corresponding section.
(d) The candidates can click on the “>” arrow which appears to the left of the question palette to
collapse the question palette thereby maximizing the question viewing window. To view the
question palette again, the candidate can click on “<” which appears on the right side of the
question window.
(e) Candidates can click on to navigate to the bottom and to navigate to the top of the question area,
without scrolling. Using the computer mouse the candidate can scroll up and down the question
viewing area for viewing the entire question.
(f) The full paper can be viewed by clicking the “Question Paper” icon on the top right corner of
the screen.
(g) Blank Sheets for doing Rough Work/calculations shall be provided to the candidates. The
Blanks Sheets would have a Header page for the candidates to write down his/her Name and
Roll Number. All calculations/writing work is to be done only in the Blank Sheets provided at
the Centre in the Examination Room/Hall and on completion of the test candidates must hand
over the rough sheets to the invigilator on duty in the Room/Hall.
(h) Navigating to a Question To navigate between questions within a Paper, the candidate
needs to do the following:

63
(a) Click on the question number in the Question Palette at the right of the screen to go to that
numbered question directly. Note that using this option does NOT save the answer to the
currently displayed question.
(b) Click on “Save and Next” to save the answer to any question. Clicking on “Save and
Next” will save the answer for the current question and the next question will be displayed
on the candidate’s computer screen.
(c) Click on “Mark for Review and Next” to mark a question for review (without answering
it) and proceed to the next question.
(i) Answering a Question
To navigate between questions within a Paper, the candidate needs to do the following:
Procedure for answering a Multiple-Choice type question:
(a) To select the option(s), click on the corresponding button(s) of the option(s).
(b) To deselect the chosen answer, click on the button of the chosen option again or click on
the “Clear Response” button.
(c) To save the answer, the candidate MUST click on the “Save and Next” button.
(d) To mark the question for review (without answering it), click on the “Mark for Review
and Next” button.
(j) Navigating through sections:
(i) Sections in the question paper are displayed on the top bar of the screen. Questions in a
section can be viewed by clicking on the section name. The section in which the candidate
is currently viewing will be highlighted.
(ii) After clicking the “Save and Next” button on the last question for a section, the candidate
will automatically be taken to the first question of the next section.
(iii) Candidates can shuffle between sections and questions within sections anytime during the
examination as per convenience only during the time stipulated.
(iv) The candidate can view the corresponding section summary as part of the legend that
appears in every section above the question palette.
(k) Procedure for answering questions that require inputs from the on-screen virtual
keyboard (numeric or otherwise):

64
(a) The candidate will have to use the on-screen virtual keyboard (that would be displayed
just below the question statement of these types of questions) and the attached computer
mouse to enter his/her answer in the space provided for the answer.
On-Screen Virtual Keyboard
(b) The answer can be changed, if required, anytime during the test. To save the answer, the candidate
MUST click on the “Save and Next” button.
(c) To mark the question for review (without answering it), click on the “Mark for Review and Next”
button.
The candidates will have the option to change previously saved answer to any question,
anytime during the entire duration of the test. To change the answer to a question that has
already been answered, first select the corresponding question from the Question Palette,
then click on “Clear Response” to clear the previously entered answer and subsequently
follow the procedure for answering that type of question.
(l) ROUGH WORK:
All calculations/writing work is to be done only in the rough sheet provided at the Centre in the
Examination Room/Hall and on completion of the test candidates must hand over the rough sheets
to the invigilator on duty in the Room/Hall.

65
Appendix-IV
NTA: Test Practice Centres (TPCs)
What is a Test Practice Centre (TPCs)?
The Ministry of Human Resource Development mandated the NTA to set up, establish, and create a
network of Test Practice Centres for candidates, especially in remote and rural areas to enable them to
practice and be comfortable in taking a Computer Based Test (CBT). This facility is completely free of
cost. Candidates can register online (on the NTA website) where they are provided a convenient TPC
near to their location to practice on a given computer node. This facilitates and eases the process of being
able to take a Computer Based Test (CBT). The entire experience of using a computer is close to the
actual experience of taking a CBT. All efforts are made to provide practice tests and questions so that
candidates can familiarize themselves with logging into the system, go through the detailed instructions
regarding the test, use the mouse or numeric keyboard on-screen (virtual) to attempt each question, scroll
down to the next question, navigate between questions, review and edit their options and submit
questions.
The objective of TPCs is primarily to organize test practice for the upcoming NTA examinations.

66
Appendix - V
Procedure to be adopted for compilation of NTA scores for multi-session Papers
(Normalization procedure based on PERCENTILE SCORE)
NTA may conduct examinations on multiple dates, generally in two sessions per day. The candidates will
be given different sets of questions per session and it is quite possible that in spite of all efforts to maintain
equivalence among various question papers, the difficulty level of these question papers administered in
different sessions may not be exactly the same. Some of the candidates may end up attempting a relatively
tougher set of questions when compared to other sets. The candidates who attempt the comparatively
tougher examination are likely to get lower marks as compared to those who attempt the easier one. In
order to overcome such a situation, a “Normalization procedure based on Percentile Score” will be
used to ensure that candidates are neither benefitted nor disadvantaged due to the difficulty level of the
examination. With the objective of ensuring that a candidate’s true merit is identified and that a level
playing field is created in the above context, the Normalization Procedure, set out below shall be adopted,
for compiling the NTA scores for multi-session papers.
The process of Normalization is an established practice for comparing candidate scores across
multi-session papers and is similar to those being adopted in other large educational selection tests
conducted in India. For normalization across sections, NTA shall use the percentile equivalence.
Percentile Scores: Percentile scores are scores based on the relative performance of all those who appear
for the examination. The marks obtained are transformed into a scale ranging from 100 to 0 for each
session of examinees.
The Percentile Score indicates the percentage of candidates that have scored EQUAL TO OR
BELOW (same or lower raw scores) that particular Percentile in that examination. Therefore the
topper (highest score) of each session will get the same Percentile of 100 which is desirable. The
marks obtained in between the highest and lowest scores are also converted to appropriate
Percentiles.
The Percentile score will be the Normalized Score for the examination (instead of the raw marks of
the candidate) and shall be used for the preparation of the merit lists.
The Percentile Scores will be calculated up to 7 decimal places to avoid the bunching effect and
reduce ties.
The Percentile score of a Candidate is calculated as follows:
100 X Number of candidates appeared in the ‘Session’ with raw score EQUAL TO OR LESS than the candidate
The total number of the candidates who appeared in the ’Session’
Note: The Percentile of the Total shall NOT be an aggregate or average of the Percentile of the individual
subject. The percentile score is not the same as the percentage of marks obtained.
Example: Suppose a test was held in 4 sessions of examinees as per details given below:
(Allocation of Days and shifts was done randomly)
(a) The distribution of candidates was as follows:
Session-1: Day-1 Shift-1, Session-2: Day-1 Shift-2, Session-3: Day-2 Shift-1 and Session-
4: Day-2 Shift-2

67
Session Day/Shift No of Candidates Marks
Absent Appeared Total Highest Lowest
Session-1 Day-1 Shift-1 3974 28012 31986 335 -39
Session-2 Day-1 Shift-2 6189 32541 38730 346 -38
Session-3 Day-2 Shift-1 6036 41326 47362 331 -49
Session-4 Day-2 Shift-2 9074 40603 49677 332 -44
Total
(Session-1 to Session-4)
25273 142482 167755 346 -49
In this method of scoring the HIGHEST RAW SCORE in each paper (irrespective of the raw scores)
will be the 100 Percentile indicating that 100% of candidates have scored equal to or lesser than the
highest scorer/ topper for that session.
Highest Raw Score and Percentile Score: All the highest raw scores will have a normalized Percentile
Score of 100 for their respective session.
Session Total
Candidate
s
Appeared
Highest
Raw
Score
Candidates
who scored
EQUAL
OR LESS
THAN
Highest
Raw Score
Percentile Score Remarks
Session-1
28012 335 28012 100.0000000
[(28012/28012)*100]
i.e. all the
highest raw
scores would
be
normalized to
100 Percentile
Score for their
respective
session.
Session -2
32541 346 32541 100.0000000
[(32541/32541)*100]
Session -3
41326 331 41326 100.0000000
[(41326/41326)*100]
Session -4
40603 332 40603 100.0000000
[(40603/40603)*100]
Lowest Raw Score and Percentile Score: The percentile Score of all the lowest raw scores will depend
on the total number of candidates who have taken the examination for their respective sessions.
68
Session Total
Candidates
Appeared
Lowest
Raw
Score
Candidates
who scored
EQUAL OR
LESS
THAN
Lowest Raw
Score
Percentile
Score
Remarks
Session -1 28012 -39 1 0.0035699
[(1/28012)*10
0]
i.e. Percentile
Score of all the
lowest raw scores
is different i.e.
Percentile Score
depends on the
total number of
candidates who
have taken the
examination for
their respective
sessions.
Session -2 32541 -38 1 0.0030730
[(1/32541)*10
0]
Session -3 41326 -49 1 0.0024198
[(1/41326)*10
0]
Session -4 40603 -44 1 0.0024629
[(1/40603)*10
0]
The following is a further explanation of the interpretation of the raw scores and Percentile Score
in Session-3 (Day-2 and Shift-1) with 41326 candidates who have taken the examination.
Candidate Percentile Score No of
Candidates
Raw
Score
Remark
A 100.0000000
[(41326/41326)*100]
1 331 This indicates that amongst those
who appeared, 100% have scored
either EQUAL TO OR LESS
THAN candidate A (331 raw
scores).
It also indicates that no candidate
has scored more than candidate A
(331 raw scores).
B 90.1224411
[(37244/41326)*100]
77 121 This indicates that amongst those
who appeared, 90.1224411%
have scored either EQUAL TO
OR LESS THAN candidate B
(121 raw scores).
It also indicates that the
remaining candidates have scored
69
more than candidate B (121 raw
scores).
C 50.4549194
[(20851/41326)*100]
381 41 This indicates that amongst those
who appeared, 50.4549194%
have scored either EQUAL TO
OR LESS THAN candidate C (41
raw scores).
It also indicates that the
remaining those who appeared
have scored more than candidate
C (41 raw scores).
D 31.7040120
[(13102/41326)*100]
789 25 This indicates that amongst those
who appeared, 31.7040120%
have scored either EQUAL TO
OR LESS THAN candidate D (25
raw scores)
It also indicates that the
remaining candidates have scored
more than candidate D (25 raw
scores).
E 1.1034216
[(456/41326)*100]
100 -15 Indicates that amongst those who
appeared, 1.1034216% have
scored either EQUAL TO OR
LESS THAN candidate E (-15
raw score)
It also indicates that the
remaining candidates have scored
more than candidate E (-15 raw
score)
STEP-BY-STEP PROCEDURE FOR NORMALIZATION AND PREPARATION OF RESULT:
Step-1: Distribution of Examinees in two shifts:
Candidates have to be distributed into two sessions randomly so that each session has an
approximately equal number of candidates. These two sessions would be as follows:
Session-1: Day-1 Shift-1, Session-2: Day-1 Shift-2
In the event of a more number of days or less number of shifts, the candidates will be divided
accordingly.

70
This will ensure that there is no bias in the distribution of candidates who shall take the examination.
Further, with a large population of examinees spread over the entire country, the possibility of such
bias becomes remote.
Step 2: Preparation of Results for each Session:
The examination results for each session will be prepared in the form of
Raw Scores
Percentiles Scores of Total raw scores.
The Percentiles would be calculated for each candidate in the Session as follows:
Let TP1 be the Percentile Score of the Total Raw Score of that candidate.
Total Percentile
(TP1) :
100 X
No. of candidates appeared from the session with raw score EQUAL TO
OR LESS than the score of the Candidate
Total No. of candidates who appeared in the session
Step-3: Compilation of NTA score and Preparation of Result:
The Percentile scores for the Total Raw Score for all the sessions (Session-1: Day-1 Shift-1,
Session-2: Day-1 Shift-2) as calculated in Step-2 above would be merged and shall be called
the NTA scores which will then be used for the compilation of results and further processing
for deciding the allocation.
In the event of the percentiles for the multi-shifts being dissimilar/unequal, the lowest will be the
eligibility cut-off for that category for all candidates (i.e. all shifts).
For Example: In the examination held in two shifts, if the 40% marks correspond to a Percentile
score of 78 in Shift 1 and 79 in Shift 2, then all those equal to or above 78 percentiles (Percentile
score of 100 to 78) in both shifts will become eligible in General Category. A similar method will
be adopted for the other categories to determine eligibility cut-offs. In case the examination is held
in more number of shifts the same principle shall apply.

71
Appendix - VI
Syllabus for JEE (Main) - 2024
Syllabus for JEE Main Paper 1 (B.E./B.Tech.)- Mathematics, Physics, and Chemistry
MATHEMATICS
UNIT 1: SETS, RELATIONS, AND FUNCTIONS:
Sets and their representation: Union, intersection, and complement of sets and their algebraic
properties; Power set; Relation, Type of relations, equivalence relations, functions; one-one,
into and onto functions, the composition of functions.
UNIT 2: COMPLEX NUMBERS AND QUADRATIC EQUATIONS:
Complex numbers as ordered pairs of reals, Representation of complex numbers in the form a
+ ib and their representation in a plane, Argand diagram, algebra of complex number, modulus,
and argument (or amplitude) of a complex number, Quadratic equations in real and complex
number system and their solutions Relations between roots and co-efficient, nature of roots,
the formation of quadratic equations with given roots.
UNIT3: MATRICES AND DETERMINANTS:
Matrices, algebra of matrices, type of matrices, determinants, and matrices of order two and
three, evaluation of determinants, area of triangles using determinants, Adjoint, and evaluation
of inverse of a square matrix using determinants and, Test of consistency and solution of
simultaneous linear equations in two or three variables using matrices.
UNIT 4: PERMUTATIONS AND COMBINATIONS:
The fundamental principle of counting, permutation as an arrangement and combination as
section, Meaning of P (n,r) and C (n,r), simple applications.
UNIT 5: BINOMIAL THEOREM AND ITS SIMPLE APPLICATIONS:
Binomial theorem for a positive integral index, general term and middle term, and simple
applications.
UNIT 6: SEQUENCE AND SERIES:
Arithmetic and Geometric progressions, insertion of arithmetic, geometric means between two
given numbers, Relation between A.M and G.M.
UNIT 7: LIMIT, CONTINUITY, AND DIFFERENTIABILITY:
Real–valued functions, algebra of functions, polynomials, rational, trigonometric, logarithmic,
and exponential functions, inverse function. Graphs of simple functions. Limits, continuity,
and differentiability. Differentiation of the sum, difference, product, and quotient of two
functions. Differentiation of trigonometric, inverse trigonometric, logarithmic, exponential,
composite, and implicit functions; derivatives of order up to two, Applications of derivatives:

72
Rate of change of quantities, monotonic-Increasing and decreasing functions, Maxima and
minima of functions of one variable,
UNIT 8: INTEGRAL CALCULAS:
Integral as an anti-derivative, Fundamental integral involving algebraic, trigonometric,
exponential, and logarithmic functions. Integrations by substitution, by parts, and by partial
functions. Integration using trigonometric identities.
Evaluation of simple integrals of the type
∫
,
∫
±
,
∫
,
∫
√
,
∫
,
∫
√
,
∫
()
,
∫
()
√
∫
±
,
∫
√
−
. The fundamental theorem of calculus, properties of definite integrals. Evaluation of definite
integrals, determining areas of the regions bounded by simple curves in standard form.
UNIT 9: DIFFRENTIAL EQUATIONS
Ordinary differential equations, their order, and degree, the solution of differential equation by
the method of separation of variables, solution of a homogeneous and linear differential
equation of the type
+
(
)
= ()
UNIT 10: CO-ORDINATE GEOMETRY
Cartesian system of rectangular coordinates in a plane, distance formula, sections formula,
locus, and its equation, the slope of a line, parallel and perpendicular lines, intercepts of a line
on the co-ordinate axis.
Straight line
Various forms of equations of a line, intersection of lines, angles between two lines, conditions
for concurrence of three lines, the distance of a point form a line, co-ordinate of the centroid,
orthocentre, and circumcentre of a triangle,
Circle, conic sections
A standard form of equations of a circle, the general form of the equation of a circle, its radius
and central, equation of a circle when the endpoints of a diameter are given, points of
intersection of a line and a circle with the centre at the origin and sections of conics, equations
of conic sections (parabola, ellipse, and hyperbola) in standard forms,
UNIT 11: THREE DIMENSIONAL GEOMETRY
Coordinates of a point in space, the distance between two points, section formula, directions
ratios, and direction cosines, and the angle between two intersecting lines. Skew lines, the
shortest distance between them, and its equation. Equations of a line

73
UNIT 12: VECTOR ALGEBRA
Vectors and scalars, the addition of vectors, components of a vector in two dimensions and
three-dimensional space, scalar and vector products,
UNIT 13: STATISTICS AND PROBABILITY
Measures of discretion; calculation of mean, median, mode of grouped and ungrouped data
calculation of standard deviation, variance, and mean deviation for grouped and ungrouped
data.
Probability: Probability of an event, addition and multiplication theorems of probability, Baye's
theorem, probability distribution of a random variate,
UNIT 14: TRIGONOMETRY
Trigonometrical identities and trigonometrical functions, inverse trigonometrical functions,
and their properties,

74
PHYSICS
UNIT 1: PHYSICS AND MEASUREMENT
Units of measurements, System of Units, S I Units, fundamental and derived units, least count,
significant figures, Errors in measurements, Dimensions of Physics quantities, dimensional
analysis, and its applications.
UNIT 2: KINEMATICS
The frame of reference, motion in a straight line, Position- time graph, speed and velocity;
Uniform and non-uniform motion, average speed and instantaneous velocity, uniformly
accelerated motion, velocity-time, position-time graph, relations for uniformly accelerated
motion, Scalars and Vectors, Vector. Addition and subtraction, scalar and vector products, Unit
Vector, Resolution of a Vector. Relative Velocity, Motion in a plane, Projectile Motion,
Uniform Circular Motion.
UNIT 3: LAWS OF MOTION
Force and inertia, Newton’s First law of motion; Momentum, Newton’s Second Law of motion,
Impulses; Newton’s Third Law of motion. Law of conservation of linear momentum and its
applications. Equilibrium of concurrent forces.
Static and Kinetic friction, laws of friction, rolling friction.
Dynamics of uniform circular motion: centripetal force and its applications: vehicle on a level
circular road, vehicle on a banked road.
UNIT 4: WORK, ENERGY, AND POWER
Work done by a constant force and a variable force; kinetic and potential energies, work-energy
theorem, power.
The potential energy of spring conservation of mechanical energy, conservative and non-
conservative forces; motion in a vertical circle: Elastic and inelastic collisions in one and two
dimensions.
UNIT5: ROTATIONAL MOTION
Centre of the mass of a two-particle system, Centre of the mass of a rigid body; Basic concepts
of rotational motion; moment of a force; torque, angular momentum, conservation of angular
momentum and its applications;
The moment of inertia, the radius of gyration, values of moments of inertia for simple
geometrical objects, parallel and perpendicular axes theorems, and their applications.
Equilibrium of rigid bodies, rigid body rotation and equations of rotational motion, comparison
of linear and rotational motions.

75
UNIT 6: GRAVITATION
The universal law of gravitation. Acceleration due to gravity and its variation with altitude and
depth. Kepler’s law of planetary motion. Gravitational potential energy; gravitational potential.
Escape velocity, Motion of a satellite, orbital velocity, time period, and energy of satellite.
UNIT 7: PROPERTIES OF SOLIDS AND LIQUIDS
Elastic behaviour, Stress-strain relationship, Hooke's Law. Young's modulus, bulk modulus,
and modulus of rigidity. Pressure due to a fluid column; Pascal's law and its applications. Effect
of gravity on fluid pressure.
Viscosity. Stokes' law. terminal velocity, streamline, and turbulent flow.critical velocity.
Bernoulli's principle and its applications.
Surface energy and surface tension, angle of contact, excess of pressure across a curved surface,
application of surface tension - drops, bubbles, and capillary rise. Heat, temperature, thermal
expansion; specific heat capacity, calorimetry; change of state, latent heat. Heat transfer-
conduction, convection, and radiation.
UNIT 8: THERMODYNAMICS
Thermal equilibrium, zeroth law of thermodynamics, the concept of temperature. Heat, work,
and internal energy. The first law of thermodynamics, isothermal and adiabatic processes.
The second law of thermodynamics: reversible and irreversible processes.
UNIT 9: KINETIC THEORY OF GASES
Equation of state of a perfect gas, work done on compressing a gas, Kinetic theory of gases -
assumptions, the concept of pressure. Kinetic interpretation of temperature: RMS speed of gas
molecules: Degrees of freedom. Law of equipartition of energy and applications to specific heat
capacities of gases; Mean free path. Avogadro's number.
UNIT 10: OSCILLATIONS AND WAVES
Oscillations and periodic motion – time period, frequency, displacement as a function of time.
Periodic functions. Simple harmonic motion (S.H.M.) and its equation; phase: oscillations of a
spring -restoring force and force constant: energy in S.H.M. - Kinetic and potential energies;
Simple pendulum - derivation of expression for its time period:
Wave motion. Longitudinal and transverse waves, speed of the travelling wave. Displacement
relation for a progressive wave. Principle of superposition of waves, reflection of waves.
Standing waves in strings and organ pipes, fundamental mode, and harmonics. Beats.
UNIT 11: ELECTROSTATICS
Electric charges: Conservation of charge. Coulomb's law forces between two point charges,
forces between multiple charges: superposition principle and continuous charge distribution.
Electric field: Electric field due to a point charge, Electric field lines. Electric dipole, Electric
field due to a dipole. Torque on a dipole in a uniform electric field.

76
Electric flux. Gauss's law and its applications to find field due to infinitely long uniformly
charged straight wire uniformly charged infinite plane sheet, and uniformly charged thin
spherical shell. Electric potential and its calculation for a point charge, electric dipole and
system of charges; potential difference, Equipotential surfaces, Electrical potential energy of
a system of two point charges and of electric dipole in an electrostatic field.
Conductors and insulators. Dielectrics and electric polarization, capacitors and capacitances,
the combination of capacitors in series and parallel, and capacitance of a parallel plate capacitor
with and without dielectric medium between the plates. Energy stored in a capacitor.
UNIT 12: CURRENT ELECTRICITY
Electric current. Drift velocity, mobility, and their relation with electric current. Ohm's law.
Electrical resistance. V-l characteristics of Ohmic and non-ohmic conductors. Electrical energy
and power. Electrical resistivity and conductivity. Series and parallel combinations of resistors;
Temperature dependence of resistance.
Internal resistance, potential difference, and emf of a cell, a combination of cells in series and
parallel. Kirchhoff’s laws and their applications. Wheatstone bridge. Metre Bridge.
UNIT 13: MAGNETIC EFFECTS OF CURRENT AND MAGNETISM
Biot - Savart law and its application to the current carrying circular loop. Ampere's law and its
applications to infinitely long current carrying straight wire and solenoid. Force on a moving
charge in uniform magnetic and electric fields.
Force on a current-carrying conductor in a uniform magnetic field. The force between two
parallel currents carrying conductors-definition of ampere. Torque experienced by a current
loop in a uniform magnetic field: Moving coil galvanometer, its sensitivity, and conversion to
ammeter and voltmeter.
Current loop as a magnetic dipole and its magnetic dipole moment. Bar magnet as an equivalent
solenoid, magnetic field lines; Magnetic field due to a magnetic dipole (bar magnet) along its
axis and perpendicular to its axis. Torque on a magnetic dipole in a uniform magnetic field.
Para-, dia- and ferromagnetic substances with examples, the effect of temperature on magnetic
properties.
UNIT 14: ELECTROMAGNETIC INDUCTION AND ALTERNATING CURRENTS
Electromagnetic induction: Faraday's law. Induced emf and current: Lenz’s Law, Eddy
currents. Self and mutual inductance. Alternating currents, peak and RMS value of alternating
current/ voltage: reactance and impedance: LCR series circuit, resonance: power in AC circuits,
wattless current. AC generator and transformer.
UNIT 15: ELECTROMAGNETIC WAVES
Displacement current. Electromagnetic waves and their characteristics, Transverse nature of
electromagnetic waves, Electromagnetic spectrum (radio waves, microwaves, infrared, visible,
ultraviolet. X-rays. Gamma rays), Applications of e.m. waves.

77
UNIT 16: OPTICS
Reflection of light, spherical mirrors, mirror formula. Refraction of light at plane and spherical
surfaces, thin lens formula, and lens maker formula. Total internal reflection and its
applications. Magnification. Power of a Lens. Combination of thin lenses in contact. Refraction
of light through a prism. Microscope and Astronomical Telescope (reflecting and refracting )
and their magnifying powers.
Wave optics: wavefront and Huygens' principle. Laws of reflection and refraction using
Huygens principle. Interference, Young's double-slit experiment, and expression for fringe
width, coherent sources, and sustained interference of light. Diffraction due to a single slit,
width of central maximum. Polarization, plane-polarized light: Brewster's law, uses of plane-
polarized light and Polaroid.
UNIT 17: DUAL NATURE OF MATTER AND RADIATION
Dual nature of radiation. Photoelectric effect. Hertz and Lenard's observations; Einstein's
photoelectric equation: particle nature of light. Matter waves-wave nature of particle, de
Broglie relation.
UNIT 18: ATOMS AND NUCLEI
Alpha-particle scattering experiment; Rutherford's model of atom; Bohr model, energy levels,
hydrogen spectrum. Composition and size of nucleus, atomic masses, Mass-energy relation,
mass defect; binding energy per nucleon and its variation with mass number, nuclear fission,
and fusion.
UNIT 19: ELECTRONIC DEVICES
Semiconductors; semiconductor diode: I-V characteristics in forward and reverse bias; diode
as a rectifier; I-V characteristics of LED. the photodiode, solar cell, and Zener diode; Zener
diode as a voltage regulator. Logic gates (OR. AND. NOT. NAND and NOR).
UNIT 20: EXPERIMENTAL SKILLS
Familiarity with the basic approach and observations of the experiments and activities:
1. Vernier calipers -its use to measure the internal and external diameter and depth of a vessel.
2. Screw gauge-its use to determine the thickness/ diameter of thin sheet/wire.
3. Simple Pendulum-dissipation of energy by plotting a graph between the square of amplitude
and time.
4. Metre Scale - the mass of a given object by the principle of moments.
5. Young's modulus of elasticity of the material of a metallic wire.
6. Surf ace tension of water by capillary rise and effect of detergents,
7. Co-efficient of Viscosity of a given viscous liquid by measuring the terminal velocity of a
given spherical body,
8. Speed of sound in air at room temperature using a resonance tube,
9. Specific heat capacity of a given (i) solid and (ii) liquid by method of mixtures.
10. The resistivity of the material of a given wire using a metre bridge.
11. The resistance of a given wire using Ohm's law.

78
12. Resistance and figure of merit of a galvanometer by half deflection method.
13. The focal length of;
(i) Convex mirror
(ii) Concave mirror, and
(ii) Convex lens, using the parallax method.
14. The plot of the angle of deviation vs angle of incidence for a triangular prism.
15. The refractive index of a glass slab using a travelling microscope.
16. Characteristic curves of a p-n junction diode in forward and reverse bias.
17. Characteristic curves of a Zener diode and finding reverse break down voltage.
18. Identification of Diode. LED,. Resistor. A capacitor from a mixed collection of such items.

79
CHEMISTRY
PHYSICAL CHEMISTRY
UNIT I: SOME BASIC CONCEPTS IN CHEMISTRY
Matter and its nature, Dalton's atomic theory: Concept of atom, molecule, element, and
compound:: Laws of chemical combination; Atomic and molecular masses, mole concept,
molar mass, percentage composition, empirical and molecular formulae: Chemical equations
and stoichiometry.
UNIT 2: ATOMIC STRUCTURE
Nature of electromagnetic radiation, photoelectric effect; Spectrum of the hydrogen atom. Bohr
model of a hydrogen atom - its postulates, derivation of the relations for the energy of the
electron and radii of the different orbits, limitations of Bohr's model; Dual nature of matter, de
Broglie's relationship. Heisenberg uncertainty principle. Elementary ideas of quantum
mechanics, quantum mechanics, the quantum mechanical model of the atom, and its important
features. Concept of atomic orbitals as one-electron wave functions: Variation of and
2
with r for 1s and 2s orbitals; various
quantum numbers (principal, angular momentum, and magnetic quantum numbers) and their
significance; shapes of s, p, and d - orbitals, electron spin, and spin quantum number: Rules
for filling electrons in orbitals – Aufbau principle. Pauli's exclusion principle and Hund's rule,
electronic configuration of elements, and extra stability of half-filled and completely filled
orbitals.
UNIT 3: CHEMICAL BONDING AND MOLECULAR STRUCTURE
Kossel-Lewis approach to chemical bond formation, the concept of ionic and covalent bonds.
Ionic Bonding: Formation of ionic bonds, factors affecting the formation of ionic bonds;
calculation of lattice enthalpy.
Covalent Bonding: Concept of electronegativity. Fajan’s rule, dipole moment: Valence Shell
Electron Pair Repulsion (VSEPR ) theory and shapes of simple molecules.
Quantum mechanical approach to covalent bonding: Valence bond theory - its important
features, the concept of hybridization involving s, p, and d orbitals; Resonance.
Molecular Orbital Theory - Its important features. LCAOs, types of molecular orbitals
(bonding, antibonding), sigma and pi-bonds, molecular orbital electronic configurations of
homonuclear diatomic molecules, the concept of bond order, bond length, and bond energy.
Elementary idea of metallic bonding. Hydrogen bonding and its applications.
UNIT 4: CHEMICAL THERMODYNAMICS
Fundamentals of thermodynamics: System and surroundings, extensive and intensive
properties, state functions, Entropy, types of processes.
The first law of thermodynamics - Concept of work, heat internal energy and enthalpy, heat
capacity, molar heat capacity; Hess’s law of constant heat summation; Enthalpies of bond

80
dissociation, combustion, formation, atomization, sublimation, phase transition, hydration,
ionization, and solution.
The second law of thermodynamics - Spontaneity of processes; S of the universe and G
of the system as criteria for spontaneity. G (Standard Gibbs energy change) and equilibrium
constant.
UNIT 5: SOLUTIONS
Different methods for expressing the concentration of solution - molality, molarity, mole
fraction, percentage (by volume and mass both), the vapour pressure of solutions and Raoult's
Law - Ideal and non-ideal solutions, vapour pressure - composition, plots for ideal and non-
ideal solutions; Colligative properties of dilute solutions - a relative lowering of vapour
pressure, depression of freezing point, the elevation of boiling point and osmotic pressure;
Determination of molecular mass using colligative properties; Abnormal value of molar mass,
van’t Hoff factor and its significance.
UNIT 6: EQUILIBRIUM
Meaning of equilibrium is the concept of dynamic equilibrium.
Equilibria involving physical processes: Solid-liquid, liquid-gas - gas and solid-gas
equilibria, Henry's law. General characteristics of equilibrium involving physical processes.
Equilibrium involving chemical processes: Law of chemical equilibrium, equilibrium
constants (K
p
and K
c
) and their significance, the significance of G and G in chemical
equilibrium, factors affecting equilibrium concentration, pressure, temperature, the effect of
catalyst; Le Chatelier’s principle.
Ionic equilibrium: Weak and strong electrolytes, ionization of electrolytes, various concepts
of acids and bases (Arrhenius. Bronsted - Lowry and Lewis) and their ionization, acid-base
equilibria (including multistage ionization) and ionization constants, ionization of water. pH
scale, common ion effect, hydrolysis of salts and pH of their solutions, the solubility of
sparingly soluble salts and solubility products, and buffer solutions.
UNIT 7: REDOX REACTIONS AND ELECTROCHEMISTRY
Electronic concepts of oxidation and reduction, redox reactions, oxidation number, rules for
assigning oxidation number, and balancing of redox reactions.
Electrolytic and metallic conduction, conductance in electrolytic solutions, molar
conductivities and their variation with concentration: Kohlrausch’s law and its applications.
Electrochemical cells - Electrolytic and Galvanic cells, different types of electrodes, electrode
potentials including standard electrode potential, half-cell and cell reactions, emf of a Galvanic
cell and its measurement: Nernst equation and its applications; Relationship between cell
potential and Gibbs' energy change: Dry cell and lead accumulator; Fuel cells.
UNIT 8: CHEMICAL KINETICS
Rate of a chemical reaction, factors affecting the rate of reactions: concentration, temperature,
pressure, and catalyst; elementary and complex reactions, order and molecularity of reactions,
rate law, rate constant and its units, differential and integral forms of zero and first-order

81
reactions, their characteristics and half-lives, the effect of temperature on the rate of reactions,
Arrhenius theory, activation energy and its calculation, collision theory of bimolecular gaseous
reactions (no derivation).
INORGANIC CHEMISTRY
UNIT 9: CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
Modem periodic law and present form of the periodic table, s, p. d and f block elements,
periodic trends in properties of elements atomic and ionic radii, ionization enthalpy, electron
gain enthalpy, valence, oxidation states, and chemical reactivity.
UNIT 10: P- BLOCK ELEMENTS
Group -13 to Group 18 Elements
General Introduction: Electronic configuration and general trends in physical and chemical
properties of elements across the periods and down the groups; unique behaviour of the first
element in each group.
UNIT 11: d - and f- BLOCK ELEMENTS
Transition Elements
General introduction, electronic configuration, occurrence and characteristics, general trends
in properties of the first-row transition elements - physical properties, ionization enthalpy,
oxidation states, atomic radii, colour, catalytic behaviour, magnetic properties, complex
formation, interstitial compounds, alloy formation; Preparation, properties, and uses of
K
2
Cr
2
O
7
, and KMnO
4
.
Inner Transition Elements
Lanthanoids - Electronic configuration, oxidation states, and lanthanoid contraction.
Actinoids - Electronic configuration and oxidation states.
UNIT 12: CO-ORDINATION COMPOUNDS
Introduction to coordination compounds. Werner's theory; ligands, coordination number,
denticity. chelation; IUPAC nomenclature of mononuclear co-ordination compounds,
isomerism; Bonding-Valence bond approach and basic ideas of Crystal field theory, colour and
magnetic properties; Importance of co-ordination compounds (in qualitative analysis,
extraction of metals and in biological systems).
ORGANIC CHEMISTRY
UNIT 13: PURIFICATION AND CHARACTERISATION OF ORGANIC COMPOUNDS
Purification - Crystallization, sublimation, distillation, differential extraction, and
chromatography - principles and their applications.
Qualitative analysis - Detection of nitrogen, sulphur, phosphorus, and halogens.

82
Quantitative analysis (basic principles only) - Estimation of carbon, hydrogen, nitrogen,
halogens, sulphur, and phosphorus.
Calculations of empirical formulae and molecular formulae: Numerical problems in organic
quantitative analysis,
UNIT 14:SOME BASIC PRINCIPLES OF ORGANIC CHEMISTRY
Tetravalency of carbon: Shapes of simple molecules - hybridization (s and p): Classification
of organic compounds based on functional groups: and those containing halogens, oxygen,
nitrogen, and sulphur; Homologous series: Isomerism - structural and stereoisomerism.
Nomenclature (Trivial and IUPAC)
Covalent bond fission - Homolytic and heterolytic: free radicals, carbocations, and carbanions;
stability of carbocations and free radicals, electrophiles, and nucleophiles.
Electronic displacement in a covalent bond
- Inductive effect, electromeric effect, resonance, and hyperconjugation.
Common types of organic reactions- Substitution, addition, elimination, and rearrangement.
UNITS 15: HYDROCARBONS
Classification, isomerism, IUPAC nomenclature, general methods of preparation, properties,
and reactions.
Alkanes - Conformations: Sawhorse and Newman projections (of ethane): Mechanism of
halogenation of alkanes.
Alkenes - Geometrical isomerism: Mechanism of electrophilic addition: addition of hydrogen,
halogens, water, hydrogen halides (Markownikoffs and peroxide effect): Ozonolysis and
polymerization.
Alkynes - Acidic character: Addition of hydrogen, halogens, water, and hydrogen halides:
Polymerization.
Aromatic hydrocarbons - Nomenclature, benzene - structure and aromaticity: Mechanism of
electrophilic substitution: halogenation, nitration.
Friedel-Craft's alkylation and acylation, directive influence of the functional group in mono-
substituted benzene.
UNIT 16: ORGANIC COMPOUNDS CONTAINING HALOGENS
General methods of preparation, properties, and reactions; Nature of C-X bond; Mechanisms
of substitution reactions.
Uses; Environmental effects of chloroform, iodoform freons, and DDT.
UNIT 17: ORGANIC COMPOUNDS CONTAINING OXYGEN
General methods of preparation, properties, reactions, and uses.
ALCOHOLS, PHENOLS, AND ETHERS

83
Alcohols: Identification of primary, secondary, and tertiary alcohols: mechanism of
dehydration.
Phenols: Acidic nature, electrophilic substitution reactions: halogenation. nitration and
sulphonation. Reimer - Tiemann reaction.
Ethers: Structure.
Aldehyde and Ketones: Nature of carbonyl group; Nucleophilic addition to >C=O group,
relative reactivities of aldehydes and ketones; Important reactions such as - Nucleophilic
addition reactions (addition of HCN. NH
3
, and its derivatives), Grignard reagent; oxidation:
reduction (Wolf Kishner and Clemmensen); the acidity of -hydrogen. aldol condensation,
Cannizzaro reaction. Haloform reaction, Chemical tests to distinguish between aldehydes and
Ketones.
Carboxylic Acids
Acidic strength and factors affecting it,
UNIT 18: ORGANIC COMPOUNDS CONTAINING NITROGEN
General methods of preparation. Properties, reactions, and uses.
Amines: Nomenclature, classification structure, basic character, and identification of
primary, secondary, and tertiary amines and their basic character.
Diazonium Salts: Importance in synthetic organic chemistry.
UNIT 19: BIOMOLECULES
General introduction and importance of biomolecules.
CARBOHYDRATES - Classification; aldoses and ketoses: monosaccharides (glucose and
fructose) and constituent monosaccharides of oligosaccharides (sucrose, lactose, and maltose).
PROTEINS - Elementary Idea of -amino acids, peptide bond, polypeptides. Proteins:
primary, secondary, tertiary, and quaternary structure (qualitative idea only), denaturation of
proteins, enzymes.
VITAMINS – Classification and functions.
NUCLEIC ACIDS – Chemical constitution of DNA and RNA.
Biological functions of nucleic acids.
Hormones (General introduction)
UNIT 20: PRINCIPLES RELATED TO PRACTICAL CHEMISTRY
Detection of extra elements (Nitrogen, Sulphur, halogens) in organic compounds; Detection of
the following functional groups; hydroxyl (alcoholic and phenolic), carbonyl (aldehyde and
ketones) carboxyl, and amino groups in organic compounds.
The chemistry involved in the preparation of the following:
Inorganic compounds; Mohr’s salt, potash alum.

84
Organic compounds: Acetanilide, p-nitro acetanilide, aniline yellow, iodoform.
The chemistry involved in the titrimetric exercises – Acids, bases, and the use of indicators,
oxalic-acid vs KMnO
4,
Mohr’s salt vs KMnO
4
Chemical principles involved in the qualitative salt analysis:
Cations – Pb
2+,
Cu
2+,
Al
3+
, Fe
3+
, Zn
2+
, Ni
2+
, Ca
2+
, Ba
2+
, Mg
2+
, NH
Anions- CO
, S
2-
,SO
,
NO3-
, NO
2-
, Cl
-
, Br
-
, I
-
( Insoluble salts excluded).
Chemical principles involved in the following experiments:
1. Enthalpy of solution of CuSO
4
2. Enthalpy of neutralization of strong acid and strong base.
3. Preparation of lyophilic and lyophobic sols.
4. Kinetic study of the reaction of iodide ions with hydrogen peroxide at room temperature.

85
Syllabus for JEE (Main) Paper 2A (B.Arch.) - Mathematics, Aptitude Test, and Drawing
Test
Part - I MATHEMATICS
UNIT 1: SETS, RELATIONS, AND FUNCTIONS:
Sets and their representation: Union, intersection, and complement of sets and their algebraic
properties; Power set; Relation, Type of relations, equivalence relations, functions; one-one, into
and onto functions, the composition of functions.
UNIT 2: COMPLEX NUMBERS AND QUADRATIC EQUATIONS:
Complex numbers as ordered pairs of reals, Representation of complex numbers in the form a +
ib and their representation in a plane, Argand diagram, algebra of complex number, modulus, and
argument (or amplitude) of a complex number, Quadratic equations in real and complex number
system and their solutions Relations between roots and co-efficient, nature of roots, the formation
of quadratic equations with given roots.
UNIT3: MATRICES AND DETERMINANTS:
Matrices, algebra of matrices, type of matrices, determinants, and matrices of order two and three,
evaluation of determinants, area of triangles using determinants, Adjoint, and evaluation of inverse
of a square matrix using determinants and, Test of consistency and solution of simultaneous linear
equations in two or three variables using matrices.
UNIT 4: PERMUTATIONS AND COMBINATIONS:
The fundamental principle of counting, permutation as an arrangement and combination as
section, Meaning of P (n,r) and C (n,r), simple applications.
UNIT 5: BINOMIAL THEOREM AND ITS SIMPLE APPLICATIONS:
Binomial theorem for a positive integral index, general term and middle term, and simple
applications.
UNIT 6: SEQUENCE AND SERIES:
Arithmetic and Geometric progressions, insertion of arithmetic, geometric means between two
given numbers, Relation between A.M and G.M.
UNIT 7: LIMIT, CONTINUITY, AND DIFFERENTIABILITY:
Real–valued functions, algebra of functions, polynomials, rational, trigonometric, logarithmic,
and exponential functions, inverse function. Graphs of simple functions. Limits, continuity, and
differentiability. Differentiation of the sum, difference, product, and quotient of two functions.
Differentiation of trigonometric, inverse trigonometric, logarithmic, exponential, composite, and
implicit functions; derivatives of order up to two, Applications of derivatives: Rate of change of
quantities, monotonic-increasing and decreasing functions, Maxima and minima of functions of
one variable,

86
UNIT 8: INTEGRAL CALCULAS:
Integral as an anti-derivative, Fundamental integral involving algebraic, trigonometric,
exponential, and logarithms functions. Integrations by substitution, by parts, and by partial
functions. Integration using trigonometric identities.
Evaluation of simple integrals of the type
∫
,
∫
±
,
∫
,
∫
√
,
∫
,
∫
√
,
∫
()
,
∫
()
√
∫
±
,
∫
√
−
. The fundamental theorem of calculus, properties of definite integrals. Evaluation of definite
integrals, determining areas of the regions bounded by simple curves in standard form.
UNIT 9: DIFFRENTIAL EQUATIONS
Ordinary differential equations, their order, and degree, the, solution of differential equation by
the method of separation of variables, solution of a homogeneous and linear differential equation
of the type
+
(
)
= ()
UNIT 10: CO-ORDINATE GEOMETRY
Cartesian system of rectangular coordinates in a plane, distance formula, sections formula, locus,
and its equation, the slope of a line, parallel and perpendicular lines, intercepts of a line on the
coordinate axis.
Straight line
Various forms of equations of a line, intersection of lines, angles between two lines, conditions
for concurrence of three lines, the distance of a point from a line, co-ordinate of the centroid,
orthocentre, and circumcentre of a triangle,
Circle, conic sections
A standard form of equations of a circle, the general form of the equation of a circle, its radius and
central, equation of a circle when the endpoints of a diameter are given, points of intersection of
a line and a circle with the centre at the origin and sections of conics, equations of conic sections
(parabola, ellipse, and hyperbola) in standard forms,
UNIT 11: THREE-DIMENSIONAL GEOMETRY
Coordinates of a point in space, the distance between two points, section formula, directions ratios,
and direction cosines, and the angle between two intersecting lines. Skew lines, the shortest
distance between them, and its equation. Equations of a line

87
UNIT 12: VECTOR ALGEBRA
Vectors and scalars, the addition of vectors, components of a vector in two dimensions and three-
dimensional space, scalar and vector products,
UNIT 13: STATISTICS AND PROBABILITY
Measures of discretion; calculation of mean, median, mode of grouped and ungrouped data
calculation of standard deviation, variance, and mean deviation for grouped and ungrouped data.
Probability: Probability of an event, addition and multiplication theorems of probability, Baye's
theorem, probability distribution of a random variate,
UNIT 14: TRIGONOMETRY
Trigonometrical identities and trigonometrical functions, inverse trigonometrical functions, and
their properties,
Part –II APTITUDE TEST
UNIT - 1 Awareness of persons. Buildings, Materials.
Objects, Texture related to Architecture and Build-environment, Visualizing three-
dimensional objects from two-dimensional drawings. Visualizing. Different sides of three-
dimensional objects. Analytical Reasoning Mental Ability (Visual. Numerical and Verbal)
UNIT – 2 Three dimensional- perception: Understanding and appreciation of scale and
proportions of objects, building forms and elements, colour texture harmony and contrast Design
and drawing of geometrical or abstract shapes and patterns in pencil. Transformation of forms
both 2D and 3D union, subtraction rotation, development of surfaces and volumes, Generation of
plans, elevations, and 3D views of objects, creating two-dimensional and three-dimensional
compositions using given shapes and forms.
Part – III DRAWING TEST
Sketching of scenes and activities from memory of urbanscape (public space, market, festivals,
street scenes, monuments, recreational spaces, etc.). landscape (riverfronts. Jungle. Gardens, trees.
Plants, etc.) and rural life.
To be conducted in a Drawing sheet.
Note: Candidates are advised to bring pencils. Own geometry box set, crasets and colour
pencils, and crayons for the Drawing Test

88
Syllabus for JEE (Main) Paper 2B (B.Planning.) - Mathematics, Aptitude Test, and
Planning
Part - I MATHEMATICS
UNIT 1: SETS, RELATIONS, AND FUNCTIONS:
Sets and their representation: Union, intersection and complement of sets and their algebraic
properties; Power set; Relation, Type of relations, equivalence relations, functions; one-one, into
and onto functions, the composition of functions.
UNIT 2: COMPLEX NUMBERS AND QUADRATIC EQUATIONS:
Complex numbers as ordered pairs of reals, Representation of complex numbers in the form a +
ib and their representation in a plane, Argand diagram, algebra of complex number, modulus, and
argument (or amplitude) of a complex number, Quadratic equations in real and complex number
system and their solutions Relations between roots and co-efficient, nature of roots, the formation
of quadratic equations with given roots.
UNIT3: MATRICES AND DETERMINANTS:
Matrices, algebra of matrices, type of matrices, determinants, and matrices of order two and three,
evaluation of determinants, area of triangles using determinants, Adjoint, and evaluation of inverse
of a square matrix using determinants and, Test of consistency and solution of simultaneous linear
equations in two or three variables using matrices.
UNIT 4: PERMUTATIONS AND COMBINATIONS:
The fundamental principle of counting, permutation as an arrangement and combination as
section, Meaning of P (n,r) and C (n,r), simple applications.
UNIT 5: BINOMIAL THEOREM AND ITS SIMPLE APPLICATIONS:
Binomial theorem for a positive integral index, general term and middle term, and simple
applications.
UNIT 6: SEQUENCE AND SERIES:
Arithmetic and Geometric progressions, insertion of arithmetic, geometric means between two
given numbers, Relation between A.M and G.M.
UNIT 7: LIMIT, CONTINUITY, AND DIFFERENTIABILITY:
Real–valued functions, algebra of functions, polynomials, rational, trigonometric, logarithmic,
and exponential functions, inverse function. Graphs of simple functions. Limits, continuity, and
differentiability. Differentiation of the sum, difference, product, and quotient of two functions.
Differentiation of trigonometric, inverse trigonometric, logarithmic, exponential, composite, and
implicit functions; derivatives of order up to two, Applications of derivatives: Rate of change of
quantities, monotonic-increasing and decreasing functions, Maxima and minima of functions of
one variable,

89
UNIT 8: INTEGRAL CALCULAS:
Integral as an anti-derivative, Fundamental integral involving algebraic, trigonometric,
exponential, and logarithmic functions. Integrations by substitution, by parts, and by partial
functions. Integration using trigonometric identities.
Evaluation of simple integrals of the type
∫
,
∫
±
,
∫
,
∫
√
,
∫
,
∫
√
,
∫
()
,
∫
()
√
∫
±
,
∫
√
−
. The fundamental theorem of calculus, properties of definite integrals. Evaluation of definite
integrals, determining areas of the regions bounded by simple curves in standard form.
UNIT 9: DIFFRENTIAL EQUATIONS
Ordinary differential equations, their order, and degree, the solution of differential equation by the
method of separation of variables, solution of a homogeneous and linear differential equation of
the type
+
(
)
= ()
UNIT 10: CO-ORDINATE GEOMETRY
Cartesian system of rectangular coordinates in a plane, distance formula, sections formula, locus,
and its equation, the slope of a line, parallel and perpendicular lines, intercepts of a line on the
coordinate axis.
Straight line
Various forms of equations of a line, intersection of lines, angles between two lines, conditions
for concurrence of three lines, the distance of a point form a line, co-ordinate of the centroid,
orthocentre, and circumcentre of a triangle,
Circle, conic sections
A standard form of equations of a circle, the general form of the equation of a circle, its radius and
central, equation of a circle when the endpoints of a diameter are given, points of intersection of
a line and a circle with the centre at the origin and sections of conics, equations of conic sections
(parabola, ellipse, and hyperbola) in standard forms,
UNIT 11: THREE-DIMENSIONAL GEOMETRY
Coordinates of a point in space, the distance between two points, section formula, directions ratios,
and direction cosines, the angle between two intersecting lines. Skew lines, the shortest distance
between them, and its equation. Equations of a line

90
UNIT 12: VECTOR ALGEBRA
Vectors and scalars, the addition of vectors, components of a vector in two dimensions and three-
dimensional space, scalar and vector products,
UNIT 13: STATISTICS AND PROBABILITY
Measures of discretion; calculation of mean, median, mode of grouped and ungrouped data
calculation of standard deviation, variance, and mean deviation for grouped and ungrouped data.
Probability: Probability of an event, addition and multiplication theorems of probability, Baye's
theorem, probability distribution of a random variate,
UNIT 14: TRIGONOMETRY
Trigonometrical identities and trigonometrical functions, inverse trigonometrical functions, and
their properties,
Part –II APTITUDE TEST
UNIT - 1 Awareness of persons. Buildings, Materials, Objects, and Textures related to
Architecture and Build-environment, Visualizing three-dimensional objects from two-
dimensional drawings. Visualizing. Different sides of three-dimensional objects. Analytical
Reasoning Mental Ability (Visual. Numerical and Verbal)
UNIT – 2 Three dimensional- perception: Understanding and appreciation of scale and
proportions of objects, building forms and elements, colour texture harmony and contrast Design
and drawing of geometrical or abstract shapes and patterns in pencil. Transformation of forms
both 2D and 3D union, subtraction rotation, development of surfaces and volumes, Generation of
plans, elevations, and 3D views of objects, creating two-dimensional and three-dimensional
compositions using given shapes and forms.
Part – III PLANNING
UNIT-1 GENERAL AWARENESS
General knowledge questions and knowledge about prominent cities, development issues,
government programs, etc.
UNIT-2 SOCIAL SCIENCES
The idea of nationalism, nationalism in India, pre-modern world, 19
th
-century global economy,
colonialism, and colonial cities, industrialization, resources, and development, types of
resources, agriculture, water, mineral resources, industries, national economy; Human
Settlements
Power-sharing, federalism, political parties, democracy, the constitution of India

91
Economic development- economic sectors, globalization, the concept of development, poverty;
Population structure, social exclusion, and inequality, urbanization, rural development, colonial
cities,
UNIT-3 THINKING SKILLS
Comprehension (unseen passage); map reading skills, scale, distance, direction, area, etc.;
critical reasoning; understanding of charts, graphs, and tables; basic concepts of statistics and
quantitative reasoning.
************
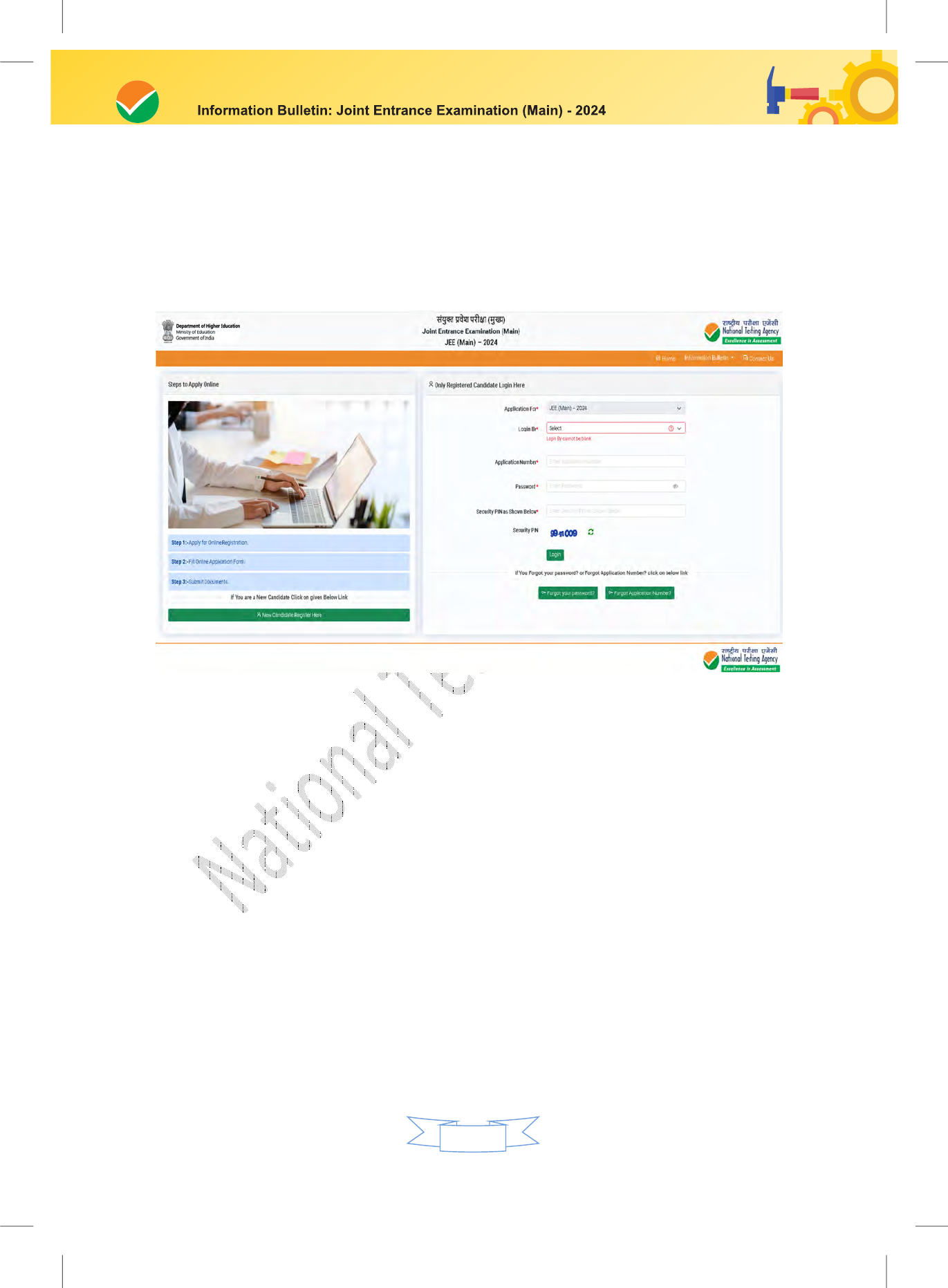
92
Appendix – VII
Replica of Online Application Form of JEE (Main)-2024 Examination

93
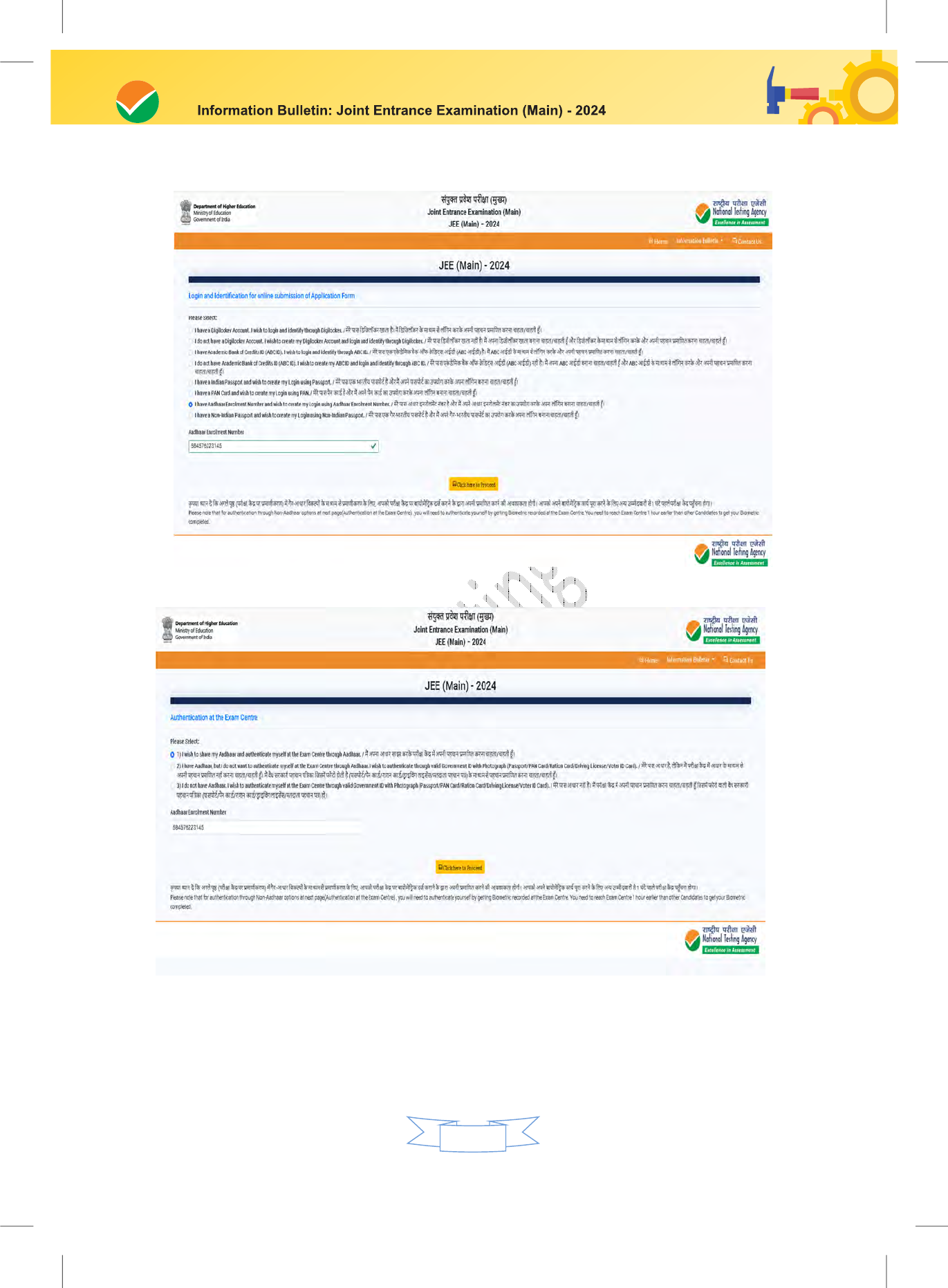
94
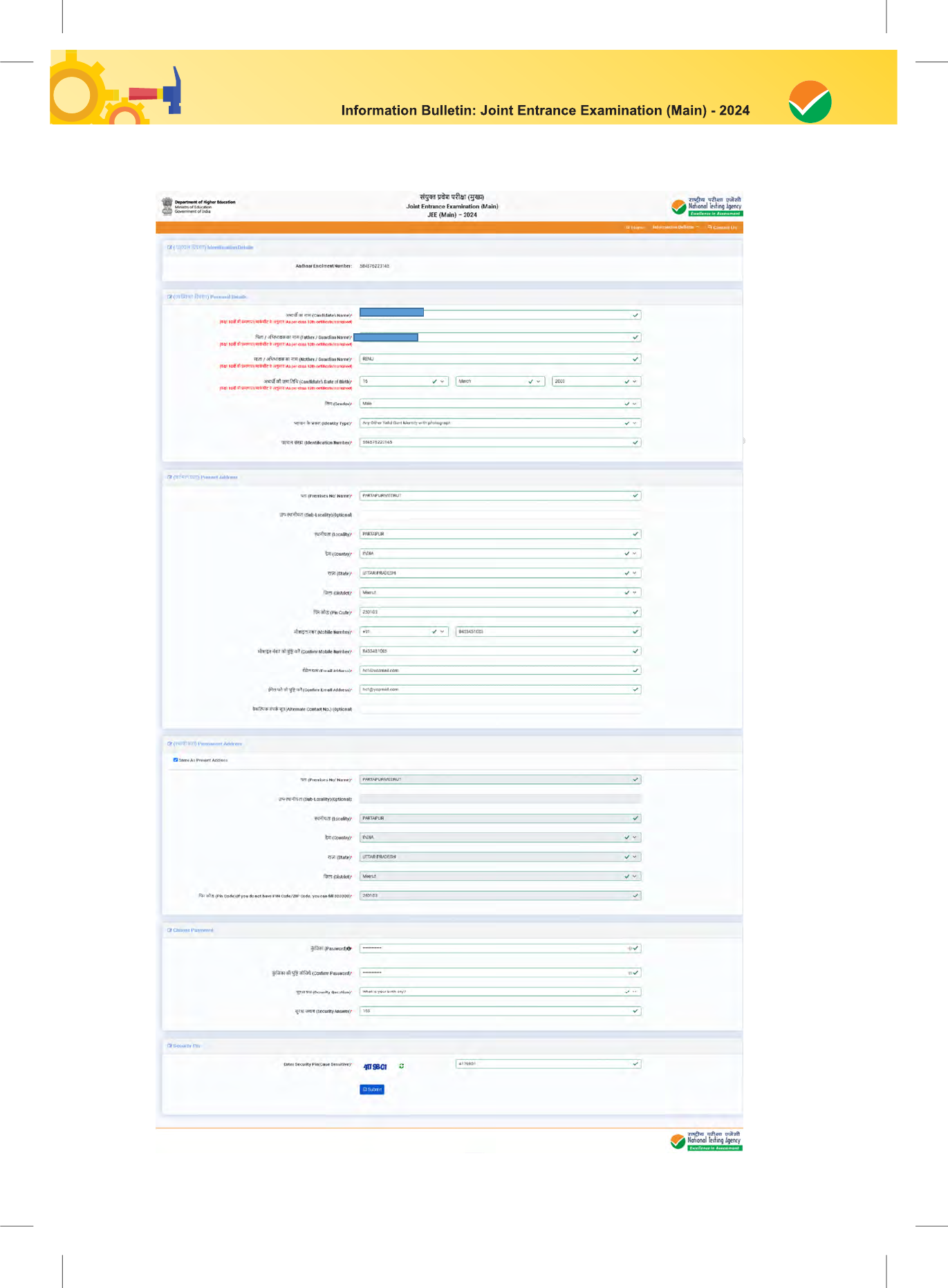
95
96
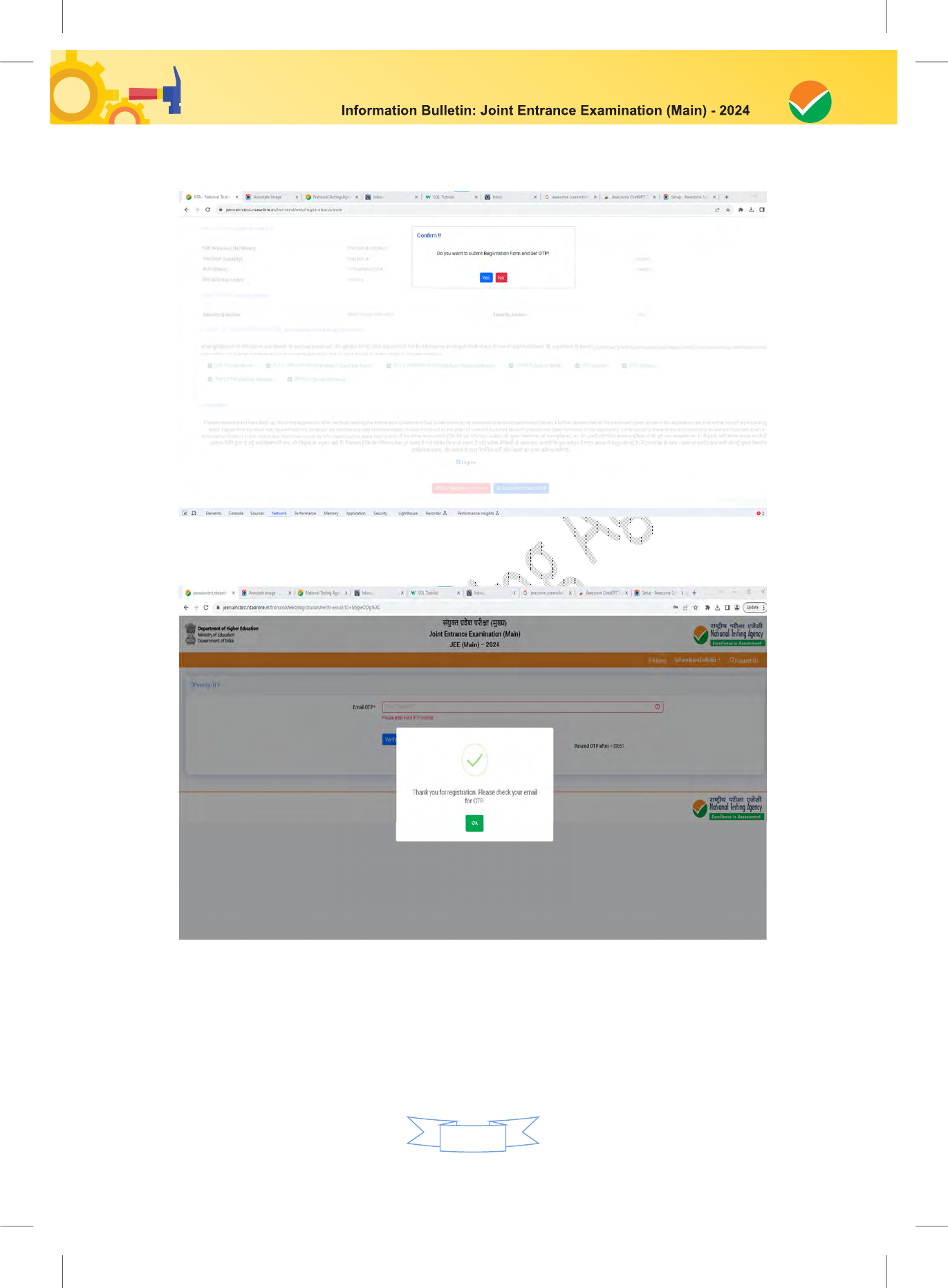
97
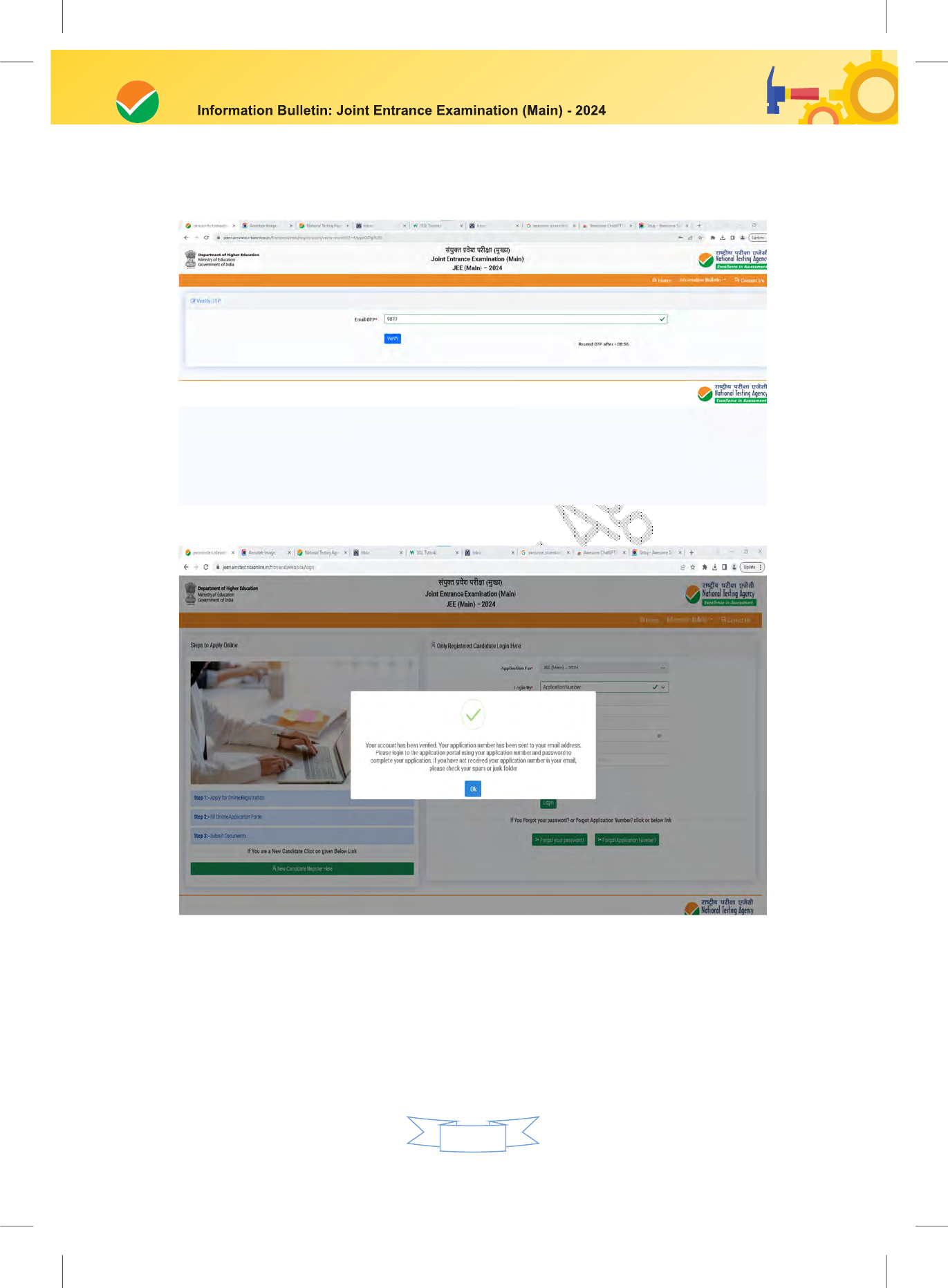
98
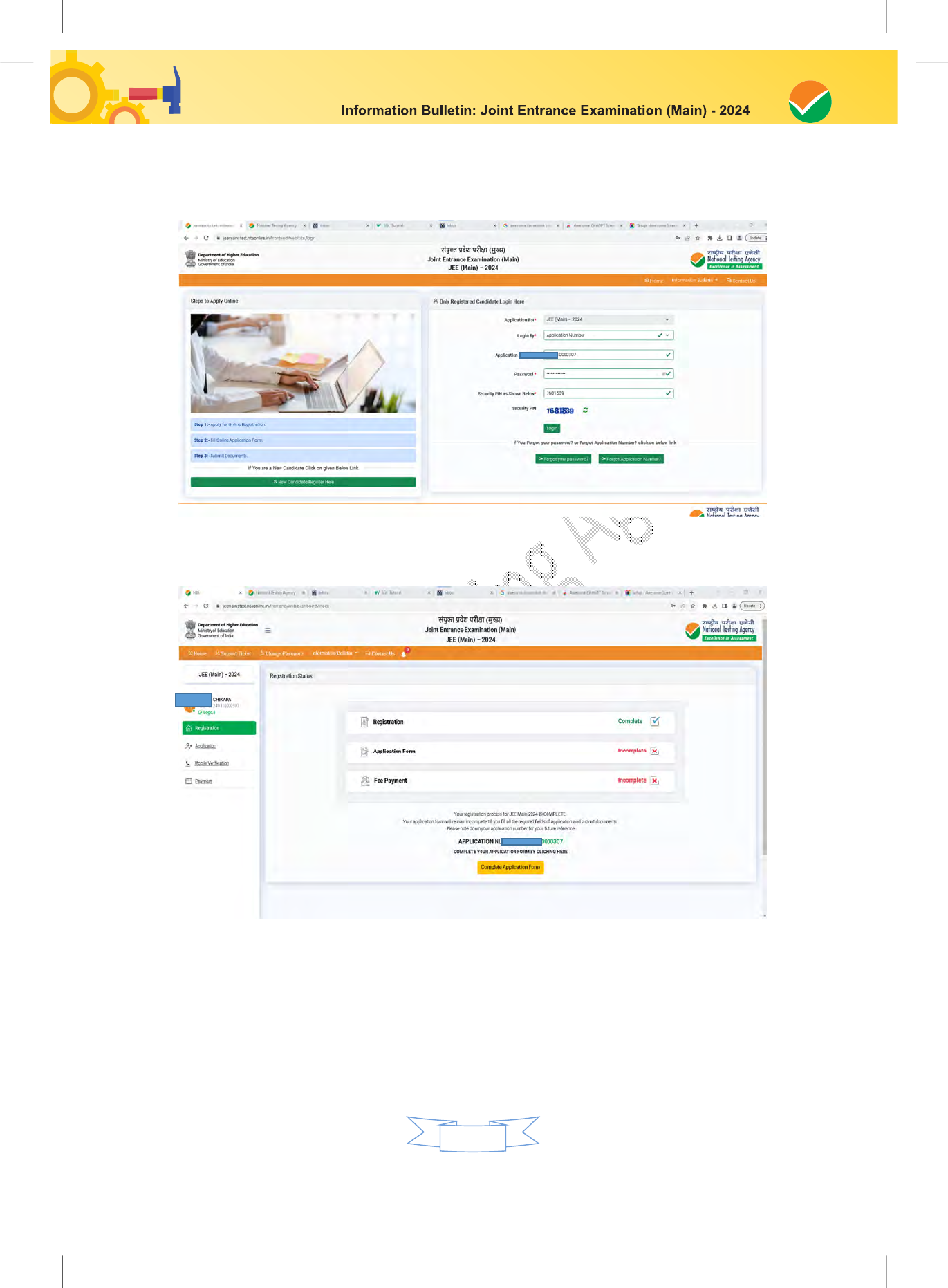
99
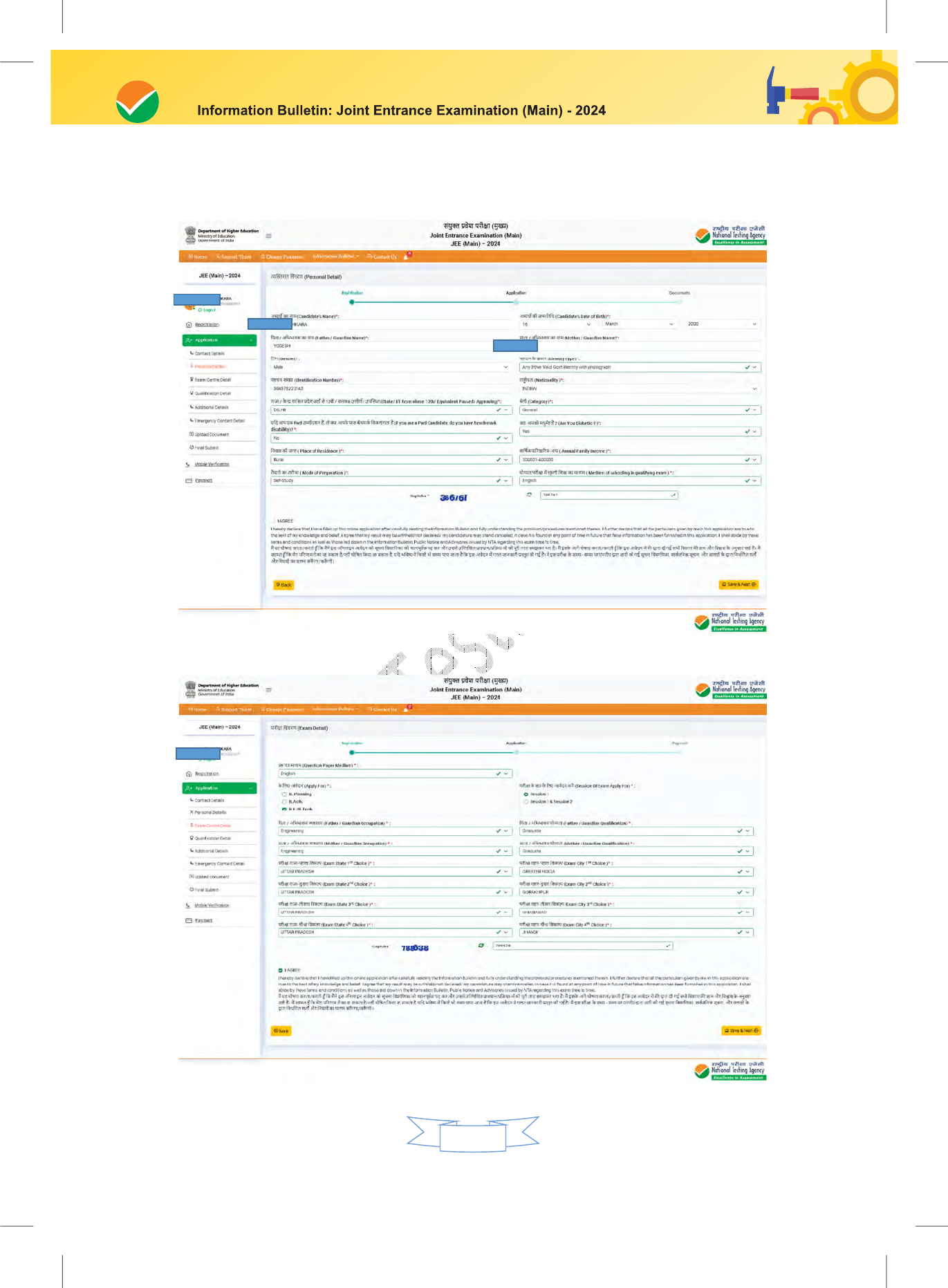
100
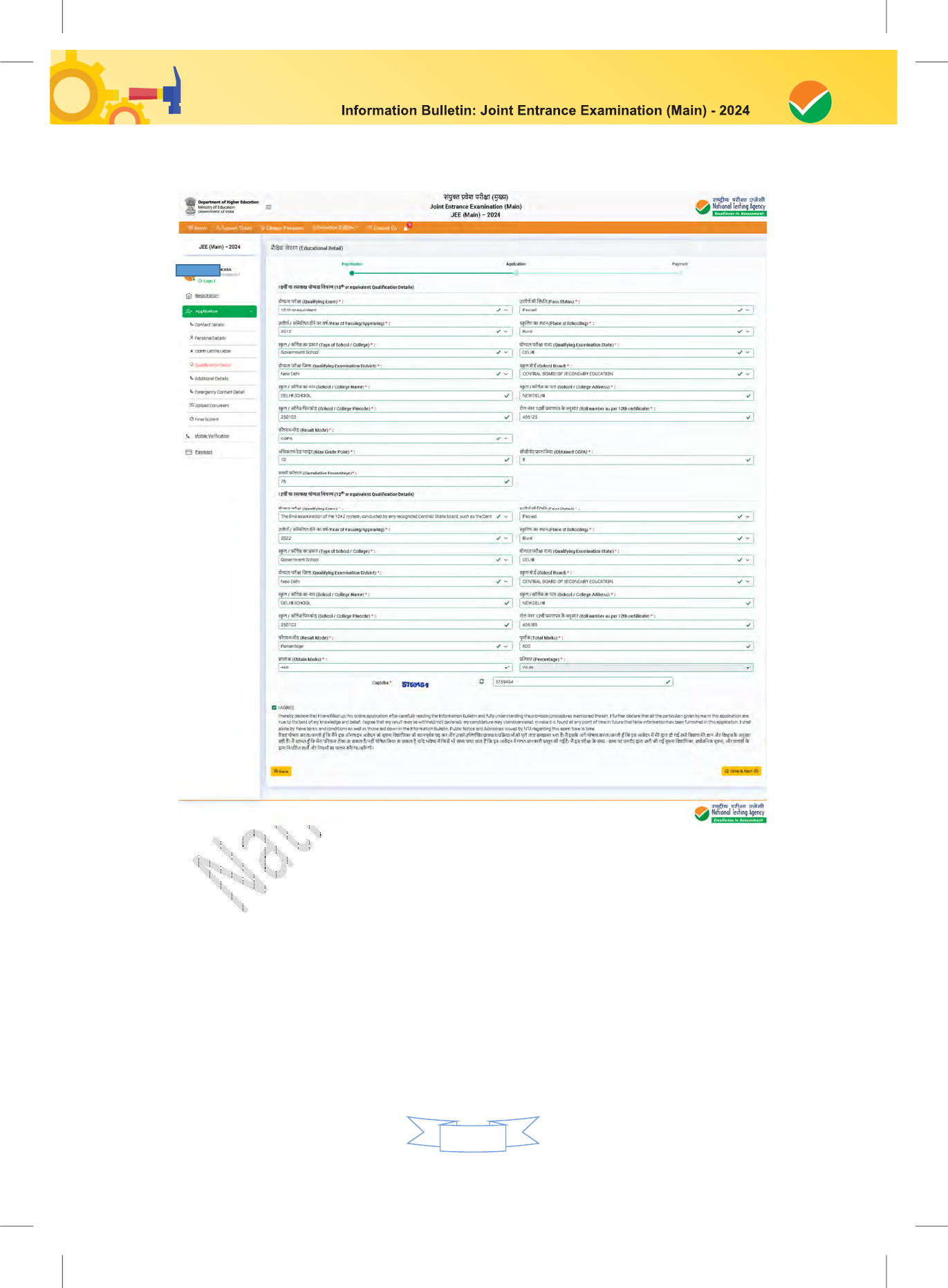
101
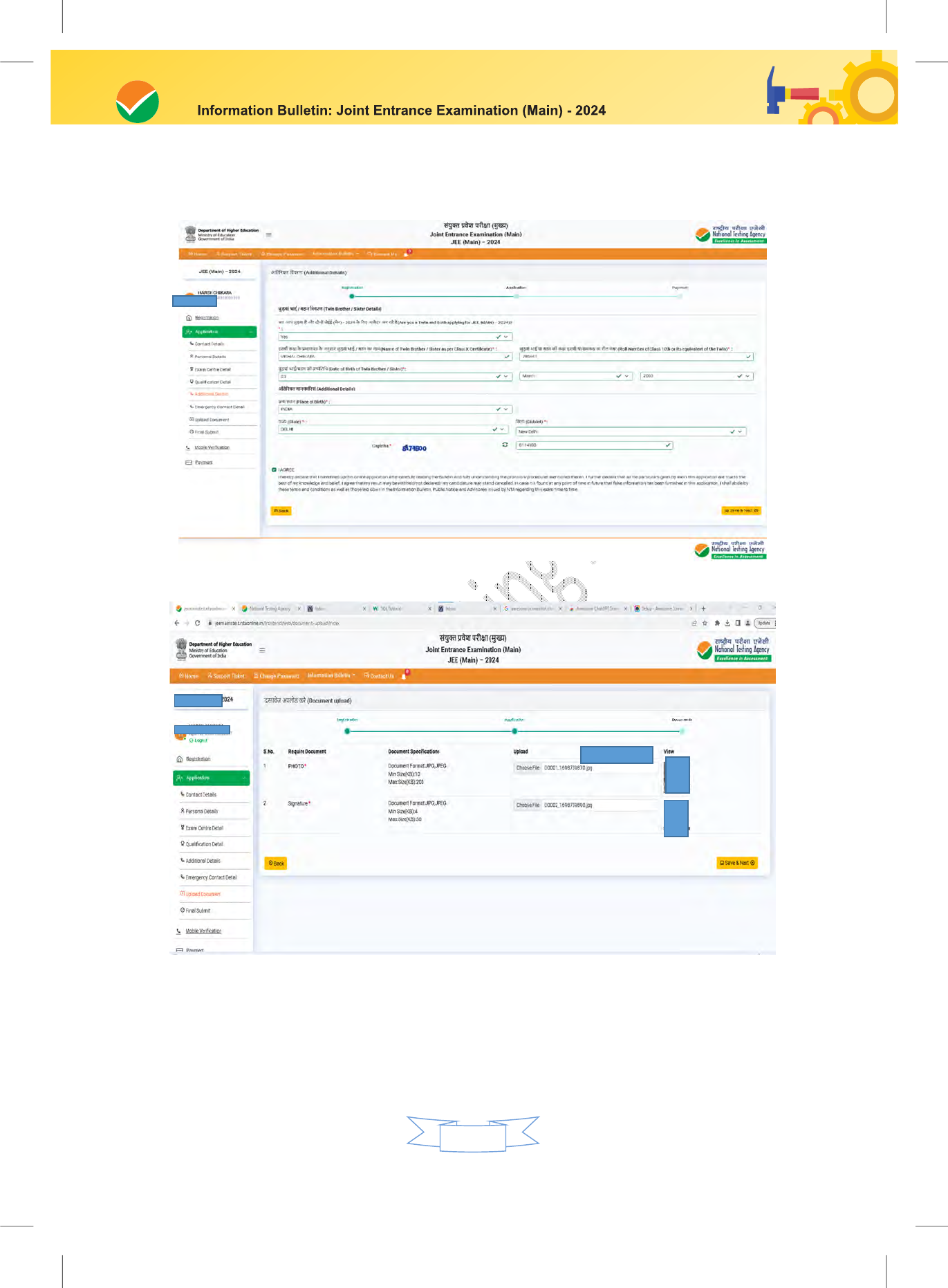
102
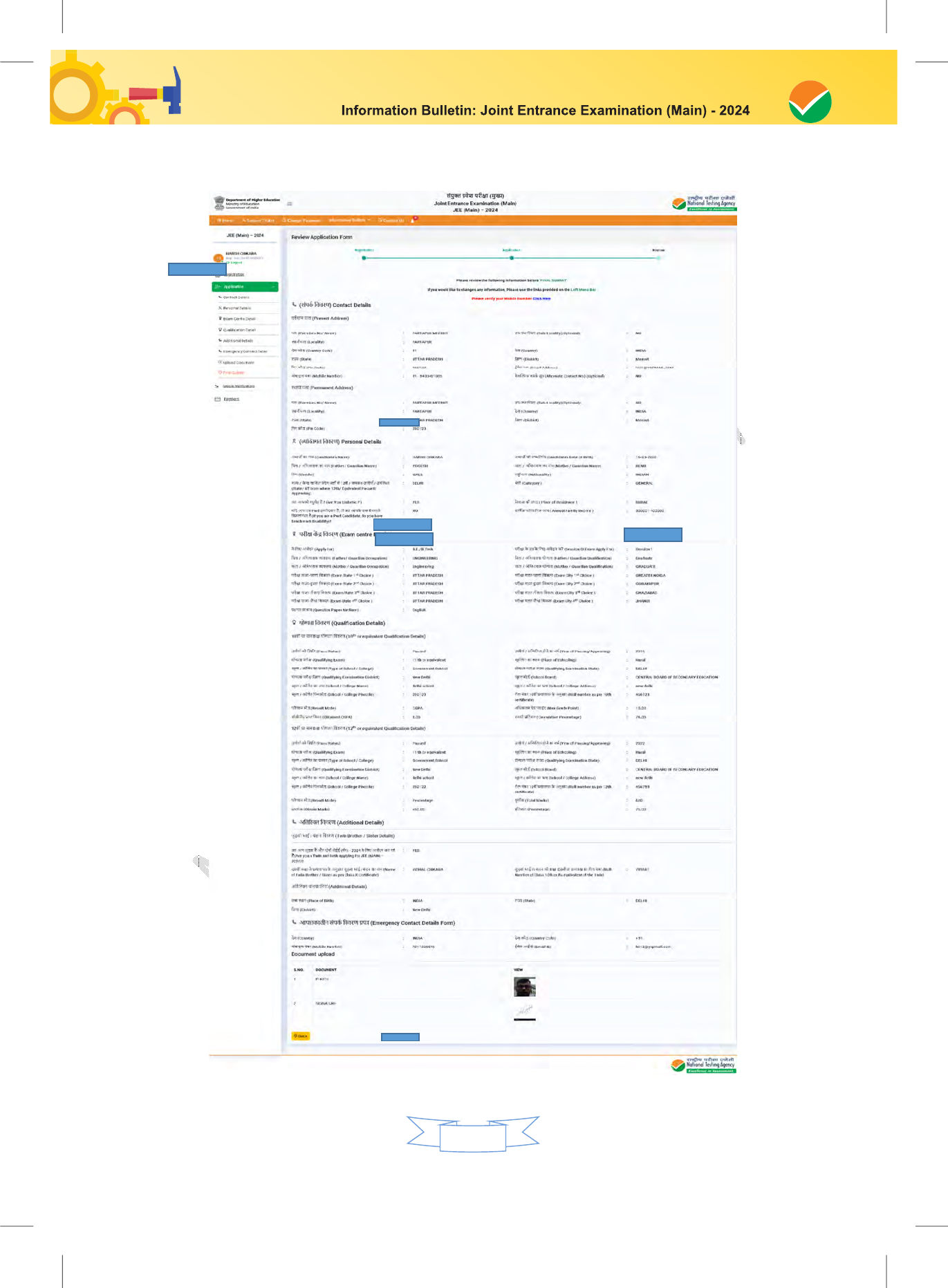
103
104
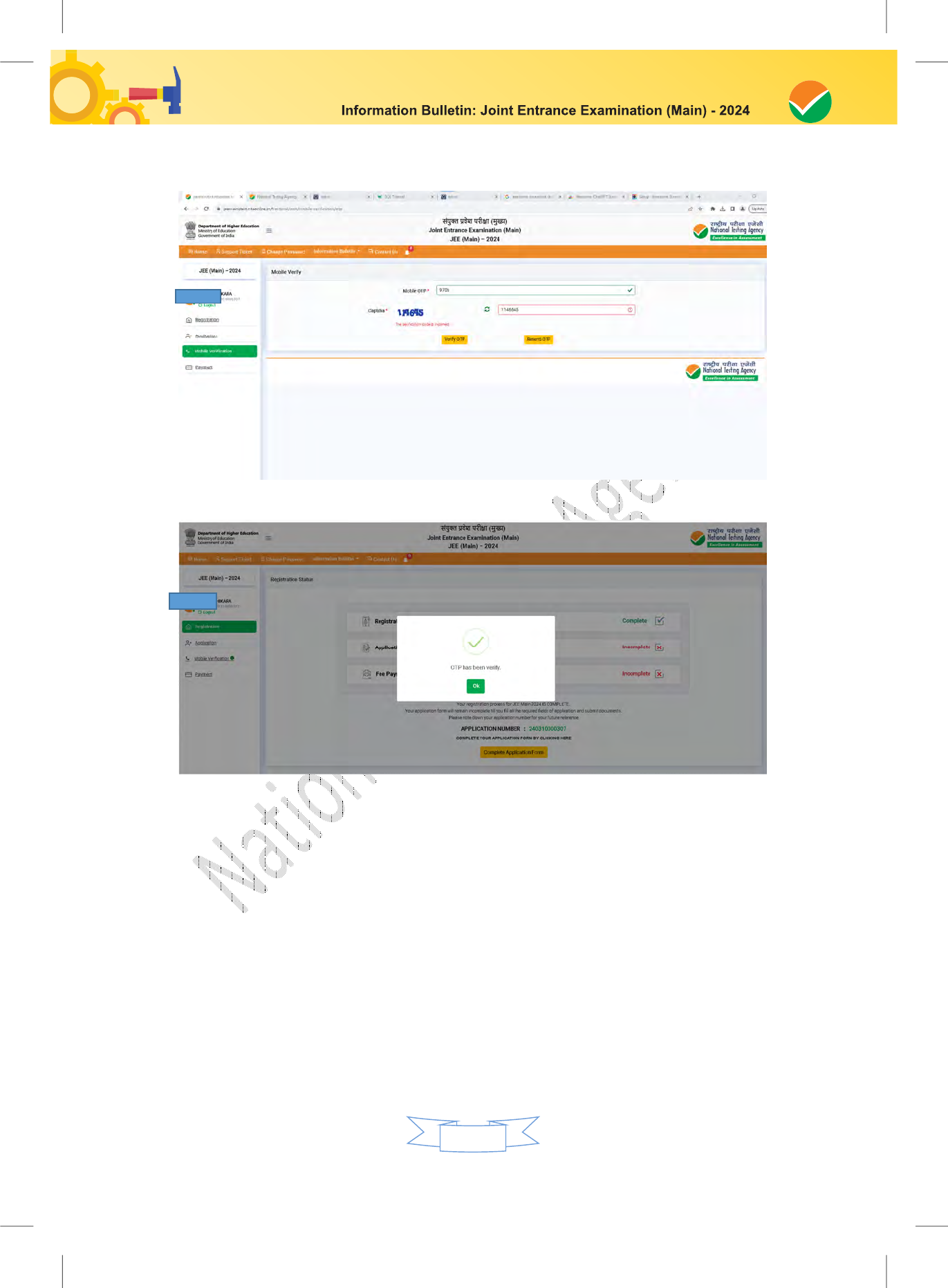
105
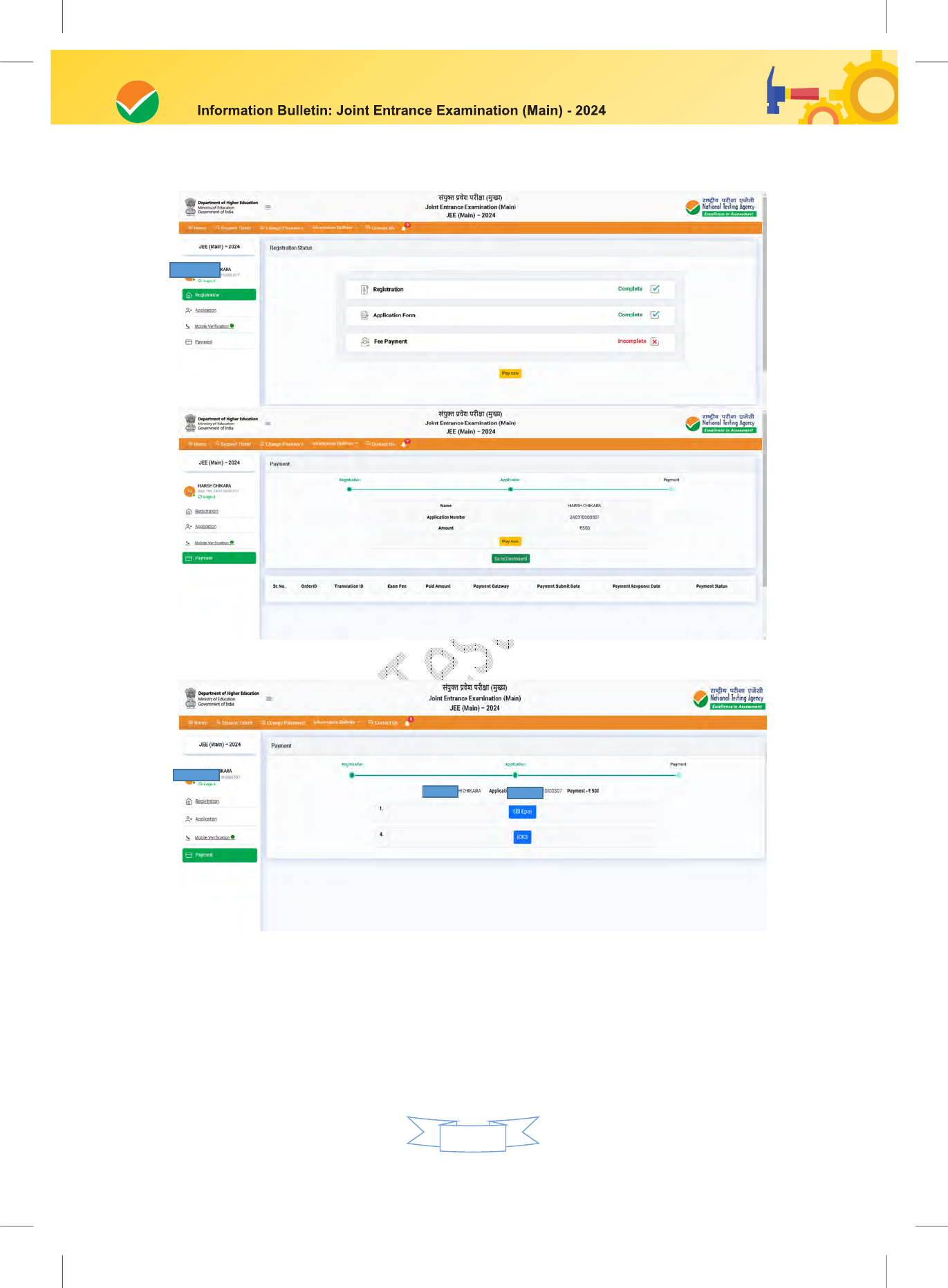
106
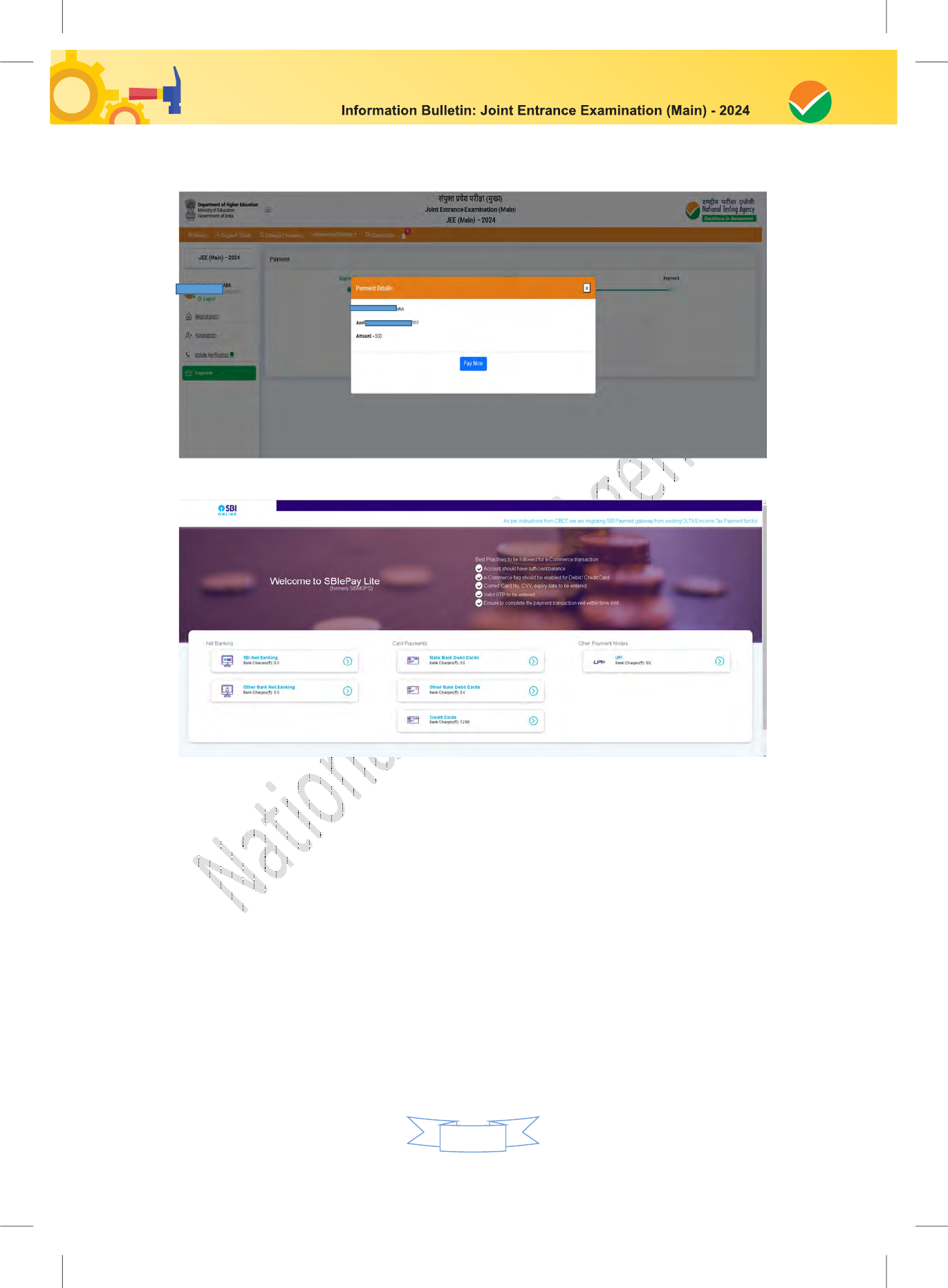
107

108

109


